
Overview
Developing high-efficiency atomic-level catalysts for energy conversion and storage technologies is crucial for addressing energy shortages. The spin states of dual-atom catalysts (DAC) are closely related to their catalytic activity. Adjusting the spin state of the active center in DAC can directly alter the occupancy of the d orbitals, thereby affecting the bonding strength between metal sites and intermediate products, as well as energy transfer.In this article, we will discuss various techniques for characterizing the spin states of atomic catalysts and strategies for modulating the spin states of their active centers. Next, we will briefly introduce studies on the spin effects in the oxygen reduction reaction (ORR), oxygen evolution reaction (OER), hydrogen evolution reaction (HER), electrocatalytic nitrogen/nitrate reduction reactions (eNRR/NO3RR), and electrocatalytic carbon dioxide reduction reactions (eCO2RR), and explain in detail how the spin regulation of DAC affects catalytic mechanisms. Finally, we will provide insights into future research directions in this critical field.
Research Background
The increasing consumption of fossil fuels and climate change severely hinder the sustainable development of human society, prompting researchers to seek effective strategies to combat these issues. Renewable energy conversion technologies involve various chemical transformation processes, providing a sustainable green route to alleviate these pressing problems. Among these technologies, electrochemical conversion processes, such as fuel cells and zinc-air batteries (involving ORR and OER), water splitting (involving OER and HER), as well as eCO2RR and eNRR/eNO3RR, can facilitate the conversion of atmospheric H2O, CO2, and N2 into high-value-added products through coupling with renewable energy, representing a promising pathway for developing sustainable energy solutions.Catalysts play a crucial role in energy conversion technologies, with over 80% of industrial processes utilizing catalysts to enhance reaction rates, efficiency, and selectivity. Currently, precious metal catalysts (such as Pt, Au, Ru, and Ir) generally outperform non-precious metal materials in facilitating reactions. However, the limited supply and high cost of precious metals significantly hinder their large-scale commercial application. Therefore, there is a need for powerful and cost-effective transition metal (TM) catalysts to help the chemical industry move towards a more sustainable future.Most catalytic processes occur on metal surfaces, leading to low catalyst utilization and limiting catalytic performance. An effective strategy to improve metal utilization is to reduce the size of the catalyst, decreasing the size of active metals in solid metal catalysts to the atomic level, forming single-atom catalysts (SAC), thereby fully utilizing metal atoms.Although SACs show great promise in the field of catalysis, these single-atom structures also have various limitations, such as low metal loading, only one specific active site, and relatively weak interactions, which hinder their development.The emergence of bimetallic nanocatalysts has facilitated the development of DACs. DACs consist of isolated pairs of metal atoms, serving as active sites. In homonuclear DACs, the same metal is present, while in heteronuclear DACs, different metals are present. Compared to SACs, DACs have many advantages, including:
- Dual-metal sites exhibit greater flexibility, allowing for the optimization of the adsorption energy of intermediate products and breaking the linear scaling relationship;
- The structure and spin states contribute to enhancing the kinetics of ORR/OER, thereby improving energy conversion efficiency and the cycle life of related energy storage devices;
- Through the interaction of electronic orbitals, DACs have greater potential to adjust the center of the d band, thereby adjusting the binding energy of intermediate products at the catalytic sites.
The initiation of chemical reactions depends on two fundamental factors: the energy involved (free energy and activation energy) and the angular momentum of the reactants (spin). The spin state (low-spin, intermediate-spin, and high-spin) or magnetic structure of the active center in DAC can directly influence the reaction energy barrier of catalytic reactions by adjusting the bonding of intermediate products. In addition to the influence of magnetism, other factors can also affect spin electrons, such as crystal field, coordination environment, oxidation state, bonding state, electron count, and conductivity.
Spin effects have been applied throughout the catalytic system; however, there has been no systematic summary reported on the spin effects in DACs.
Highlights of the Article
This review focuses on recent advances in discovering the effects of electronic spin on HER, OER, ORR, CO2RR, and eNRR/eNO3RR electrocatalysis. The main timeline of developments in DACs is highlighted in Figure 1. Various techniques for characterizing the spin states of atomic catalysts and strategies for modulating their active center spin states are discussed.
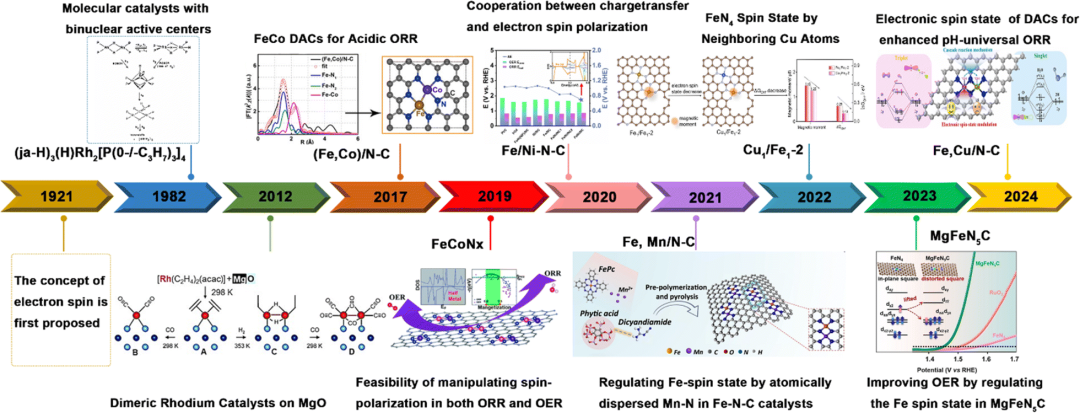
- Fig. 1 History of DACs with spin regulation for electrocatalysis.
Illustrative Analysis
1. Basic Theory of Spin
Electronic spin is an important fundamental property generated by its intrinsic angular momentum, playing a crucial role in manipulating various aspects of catalytic reactions. In the context of quantum mechanics, the co-localization of two electrons in the same orbital is only possible when they possess opposite energies. The spin state of electrons in atomic or molecular orbitals is referred to as the spin state. After interaction, the five sub-d orbitals split into low-energy t2g bonding orbitals, including dxy, dyz, and dxz, and high-energy eg antibonding orbitals, including dx2−y2 and dz2, as shown in Figure 2. Since the specific mechanisms of spin effects in different electrochemical reactions vary, understanding the spin of metals is crucial for designing DACs with stronger electrocatalytic activity. This section first introduces the descriptors of spin effects in atomic-level catalysts, catalytic mechanisms, and methods for regulating spin polarization.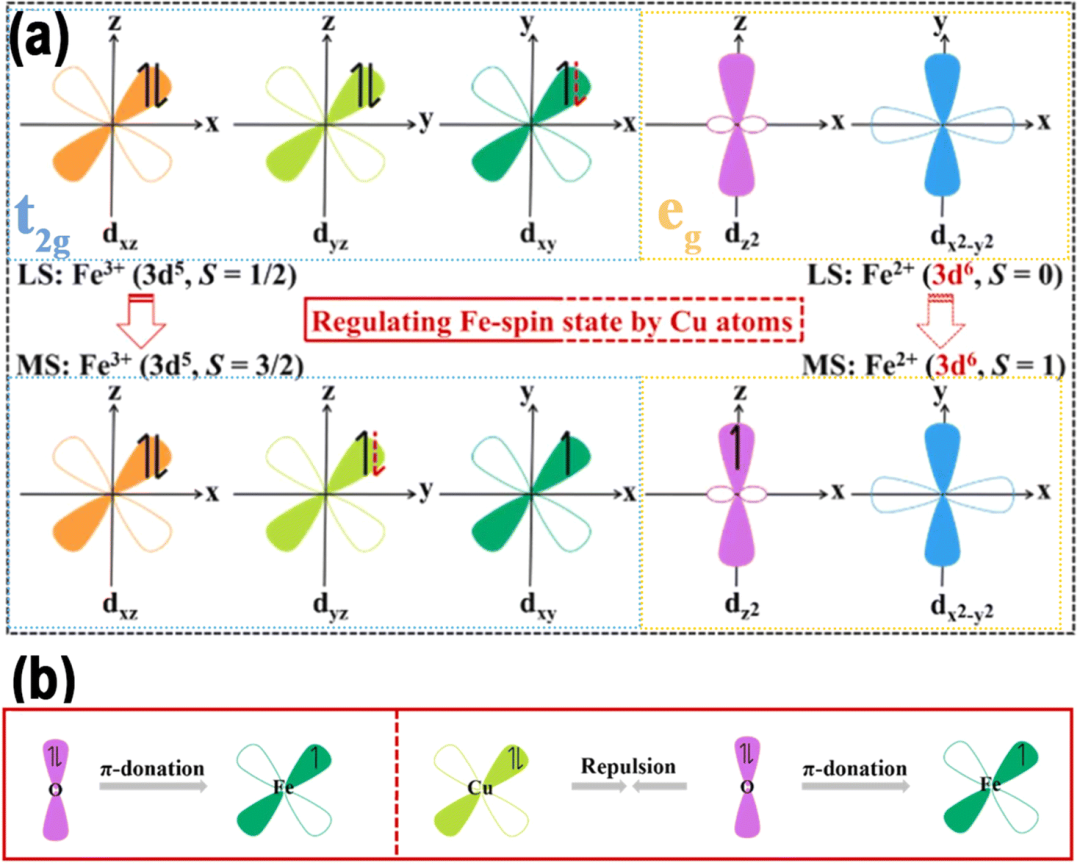
- Fig. 2 (a–c) Typical structures of SACs, DACs, and TACs.
2. Spin Effects in Dual-Atom ElectrocatalysisRecently, there has been increasing recognition that the spin states of DACs play a key role in determining their catalytic activity for HER, CO2RR, NRR, ORR, and OER. Modulating the spin state provides the possibility for accurately designing DACs with optimized electronic structures.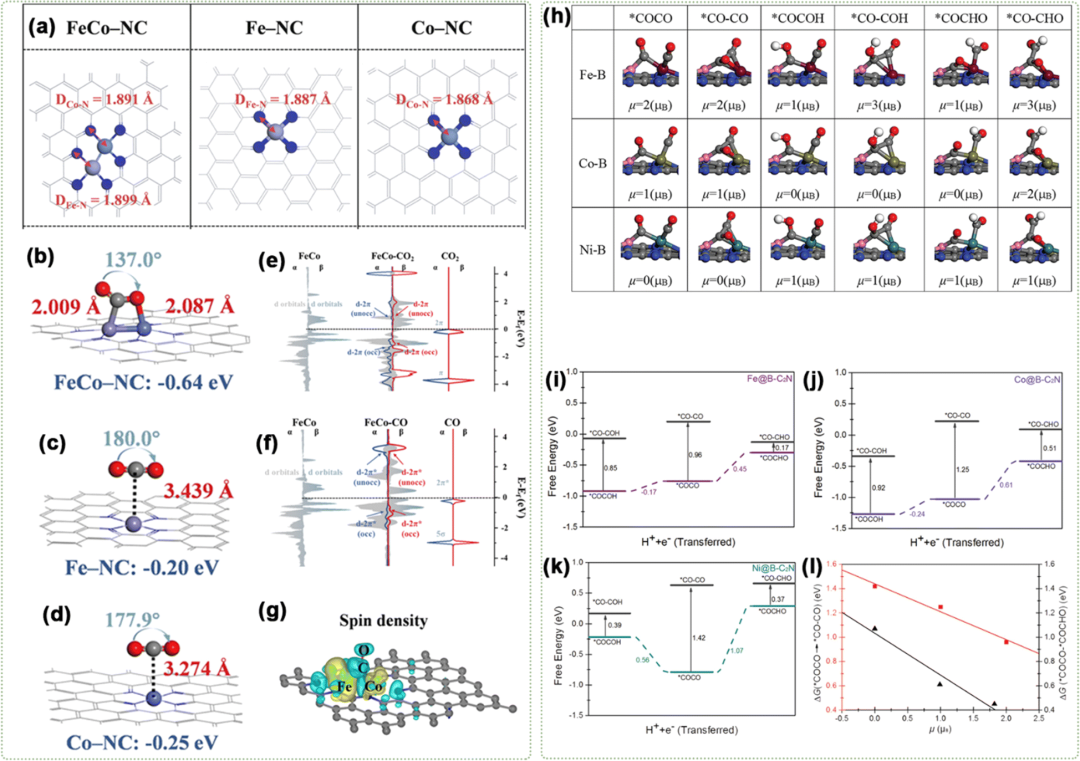
- Fig. 3 (a) Optimized structural models. Predominant configurations of CO2 adsorption for (b) FeCo–NC, (c) Fe–NC, and (d) Co–NC. Projected density of state (PDOS) analysis of (e) the 3d orbitals of Fe and Co dual-atom centers and the 2p orbitals of CO2; (f) the 3d orbitals of Fe, Co dual-atom centers, 2p orbitals of the CO gas molecule. (g) Spin density of CO-assisted FeCo–NC. (h) Optimized structure of C–C coupling. Free energy profiles for C–C coupling on (i) Fe@B-C2N, (j) Co@B-C2N, and (k) Ni@B-C2N at 0 V (vs. RHE). (l) Correlation of the reaction free energy as a function of the spin moment (m) for adsorbed reactants.
Conclusion and Outlook
Controlling the spin state of the active center in DACs has been a focal point of interest. In this review, we have discussed the impact of spin state transitions on various electrocatalytic reactions. We described the characteristics of spin state transitions and the descriptors of atomic catalysts, and then proposed strategies for modulating the spin states of DAC active centers. Finally, we summarized recent studies on the spin effects of spin DACs in ORR, OER, HER, eCO2RR, and eNRR, laying the foundation for future research.Despite the progress made, research on spin state transitions is still in its early stages. Several key issues remain worthy of further attention, including:
- The effects of various spin states in DACs on different catalytic reactions have not been fully studied. Research on the impact of catalytic reactions is still insufficient. It is currently unclear whether spin state transitions will alter the adsorption configurations and reaction pathways of intermediate products. Future studies need to integrate more advanced characterization techniques to further explore their mechanisms.
- Currently, the main methods for studying spin state transitions in DACs rely on in situ characterization techniques. During the catalytic process, the reaction intermediates adsorbed on the active sites are dynamically changing. In situ techniques cannot capture the spin state transitions of the magnetic properties of metal active centers during the reaction process. In situ characterization methods for detecting the spin states of active centers mainly include in situ Mössbauer spectroscopy and in situ X-ray emission spectroscopy (XES). Advancing in situ characterization techniques, such as in situ X-ray absorption spectroscopy (XAS) and in situ vibrating sample magnetometry (VSM), is crucial for monitoring the spin changes of active sites and reaction intermediates during catalytic reactions.
- The methods for modulating the spin states of DAC active centers are quite limited. Additionally, there is a lack of real-time control strategies for spin states during reactions. Real-time adjustment of spin states during reactions can help optimize the adsorption and desorption of key reaction intermediates, thereby enhancing the functionality of the catalysts.
- There are few reports on the spin-catalyzed mechanisms of HER, ENRR, and eCO2RR in DACs. Furthermore, understanding how changes in the spin states of their active centers affect their catalytic performance is crucial for developing more reliable electrocatalysts. Utilizing computational chemistry for theoretical calculations can not only assist experimental studies in identifying catalytic active sites but also play an important role in elucidating reaction mechanisms.
This work, titled “Spin effect in dual-atom catalysts for electrocatalysis,” was published in the journal Chemical Science by the Royal Society of Chemistry.

Paper Information
- Spin effect in dual-atom catalysts for electrocatalysisXiaoqin Xua and Jingqi Guan*
Chem. Sci., 2024, 15, 14585-14607 https://doi.org/10.1039/D4SC04370G
Author Information
 Xiaoqin Xu PhD StudentJilin University
Xiaoqin Xu PhD StudentJilin University
This article’s first author graduated with a bachelor’s degree from Jilin Agricultural University and a master’s degree from Heilongjiang University. Currently, he is a PhD student at Jilin University, under the supervision of Professor Guan Jingqi. His main research direction is the synthesis of mono- and multi-nuclear catalysts for energy conversion and storage. He has published 5 academic papers as the first/corresponding author in journals such as Adv. Mater., Chem. Sci., Appl. Catal. B Environ., Chem. Eng. J.
 Jingqi Guan ProfessorJilin UniversityThis article’s corresponding author is a “Tang Aoqing Scholar” at Jilin University, a professor, and a doctoral supervisor engaged in the synthesis of nano-cluster and single-atom materials, electrocatalysis, renewable energy, and biosensing research. He obtained his bachelor’s degree in chemistry and his PhD in science from Jilin University in 2002 and 2007, respectively. From 2012 to 2018, he conducted postdoctoral research at the University of California, Berkeley, and the Dalian Institute of Chemical Physics. To date, he has published over 210 SCI papers, including more than 180 related SCI papers as the first or corresponding author in journals such as Nat. Catal., Adv. Funct. Mater., Chem. Sci., Nano Energy, and 2 academic book chapters, with an H-index of 48. He currently serves as a young editorial board member for the journals Journal of Catalysis, Physical Chemistry Chemical Physics, and EcoEnergy.
Jingqi Guan ProfessorJilin UniversityThis article’s corresponding author is a “Tang Aoqing Scholar” at Jilin University, a professor, and a doctoral supervisor engaged in the synthesis of nano-cluster and single-atom materials, electrocatalysis, renewable energy, and biosensing research. He obtained his bachelor’s degree in chemistry and his PhD in science from Jilin University in 2002 and 2007, respectively. From 2012 to 2018, he conducted postdoctoral research at the University of California, Berkeley, and the Dalian Institute of Chemical Physics. To date, he has published over 210 SCI papers, including more than 180 related SCI papers as the first or corresponding author in journals such as Nat. Catal., Adv. Funct. Mater., Chem. Sci., Nano Energy, and 2 academic book chapters, with an H-index of 48. He currently serves as a young editorial board member for the journals Journal of Catalysis, Physical Chemistry Chemical Physics, and EcoEnergy.
- Personal Homepage:https://teachers.jlu.edu.cn/Guan_Jingqi/zh_CN/index/39768/list/index.htm
Journal Introduction


rsc.li/chemical-science
Chem. Sci.
| 2-Year Impact Factor* | 7.6 points |
| 5-Year Impact Factor* | 8.0 points |
| JCR Quartile* | Q1 Chemistry – Multidisciplinary |
| CiteScore† | 14.4 points |
| Median Peer Review Time‡ | 33 days |
Chemical Science is a multidisciplinary journal covering all areas of chemical science and is the flagship journal of the Royal Society of Chemistry. The papers published must not only be of significant importance in their respective fields but also engage a broad interest from readers in other areas of chemical science. The published papers should contain significant advancements, conceptual innovations, or profound insights into the development of the field. The scope of publication includes but is not limited to organic chemistry, inorganic chemistry, physical chemistry, materials science, nanoscience, catalysis, chemical biology, analytical chemistry, supramolecular chemistry, theoretical chemistry, computational chemistry, green chemistry, and energy and environmental chemistry. As a diamond open access journal, readers can freely access the full text of published papers, while the publication fees are covered by the Royal Society of Chemistry, and authors do not need to pay.
Editor-in-Chief
-
Andrew Cooper🇬🇧 University of Liverpool
Associate Editors
|
|
* 2023 Journal Citation Reports (Clarivate, 2024)
† CiteScore 2023 by Elsevier
‡ Median, only counting manuscripts that entered the peer review stage

