
Green synthesis of nitrates (NO3−) through the electrocatalytic nitrogen oxidation reaction (NOR) is a promising artificial nitrogen fixation strategy, showing significant advantages over traditional nitrate synthesis methods based on the Haber-Bosch process and the Oswald process. However, the poor absorbability of nitrogen, the high bond energy of N≡N (941 kJ·mol−1), and the competing multi-electron transfer oxygen evolution reaction (OER) limit the activity and selectivity of this process.
In this study, we prepared a copper nanowire (Cu-NWs) electrode anchored with irregular TiO2−x nanoparticles derived from MXene for efficient electrocatalytic nitrogen oxidation. The electrode exhibited a nitrate yield of 62.50 μg·h−1·mgcat−1 and a Faradaic efficiency (FE) of 22.04% without vacuum assistance, while the nitrate yield significantly increased to 92.63 μg·h−1·mgcat−1 with vacuum assistance, achieving a Faradaic efficiency of 40.58%. Under optimal experimental conditions, the TiO2−x/Cu-NWs electrode also showed excellent reproducibility and stability. Additionally, we assembled a Zn-N2 reaction device with TiO2−x/Cu-NWs as the anode and a zinc plate as the cathode, achieving an extremely high nitrate yield of 156.25 µg·h−1·mgcat−1. The open-circuit voltage (OCV) of this zinc-nitrate battery was 1.35 V. This study provides a new strategy to enhance the performance of N2 oxidation under environmental conditions to achieve higher nitrate yields.

1. Importance of Nitrogen Oxidation Reaction
-
The electrocatalytic nitrogen oxidation reaction (NOR) is a key step in synthesizing nitrates (NO3−), which is significant for artificial nitrogen fixation.
-
Traditional nitrate synthesis methods face issues such as high energy consumption and environmental pollution, making it crucial to find new synthesis pathways.
2. Research Motivation
-
The nonpolar characteristics and high stability of nitrogen (N2) result in poor absorbability, limiting the efficiency of the electrocatalytic nitrogen oxidation reaction.
-
This study aims to improve the absorption and conversion efficiency of nitrogen in the electrocatalytic process by designing novel electrocatalysts.
2. Research Methods
1. Catalyst Preparation
-
MXene was used as the substrate material, and TiO2−x composites were prepared through chemical etching and thermal treatment.
-
Copper nanowires (CuNWs) were uniformly deposited on the TiO2−x surface to form the CuNWs@TiO2−x composite catalyst.
2. Electrocatalytic Performance Testing
-
Under vacuum-assisted conditions, the CuNWs@TiO2−x composite catalyst was tested for electrocatalytic nitrogen oxidation using an electrochemical workstation.
-
Methods such as cyclic voltammetry, linear sweep voltammetry, and chronoamperometry were used to evaluate the catalytic performance of the catalyst.

3. Research Results
1. Enhancement of Catalytic Performance
-
The CuNWs@TiO2−x composite catalyst exhibited significant enhancement in electrocatalytic nitrogen oxidation performance under vacuum assistance.
-
Compared to single catalysts, the composite catalyst showed significant increases in nitrate yield and current density.
2. Investigation of Reaction Mechanism
-
In situ infrared spectroscopy and electrochemical impedance spectroscopy revealed the reaction mechanism of the CuNWs@TiO2−x composite catalyst in the electrocatalytic nitrogen oxidation process.
-
The study found that vacuum-assisted conditions promoted the adsorption and conversion of nitrogen on the catalyst surface, thereby improving catalytic efficiency.
4. Illustrated Guide
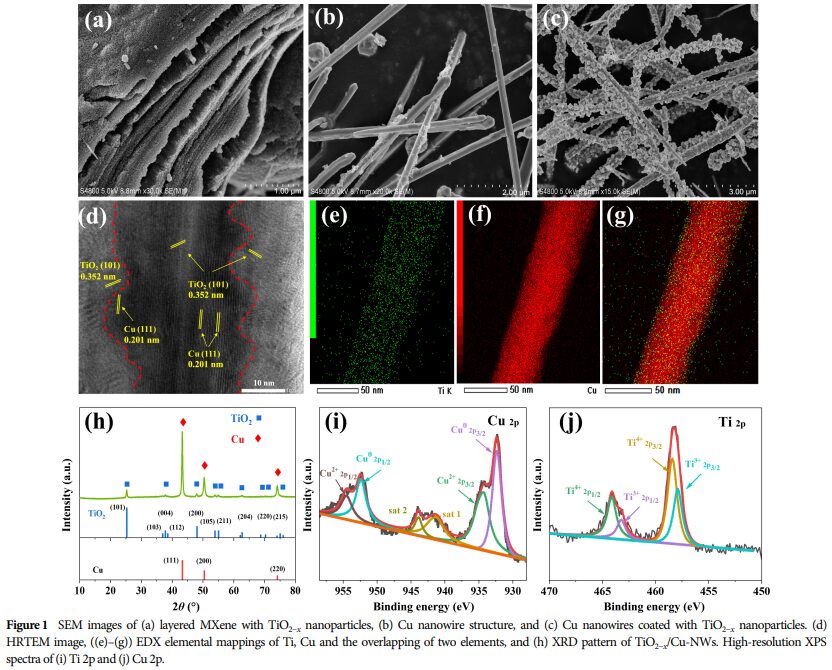
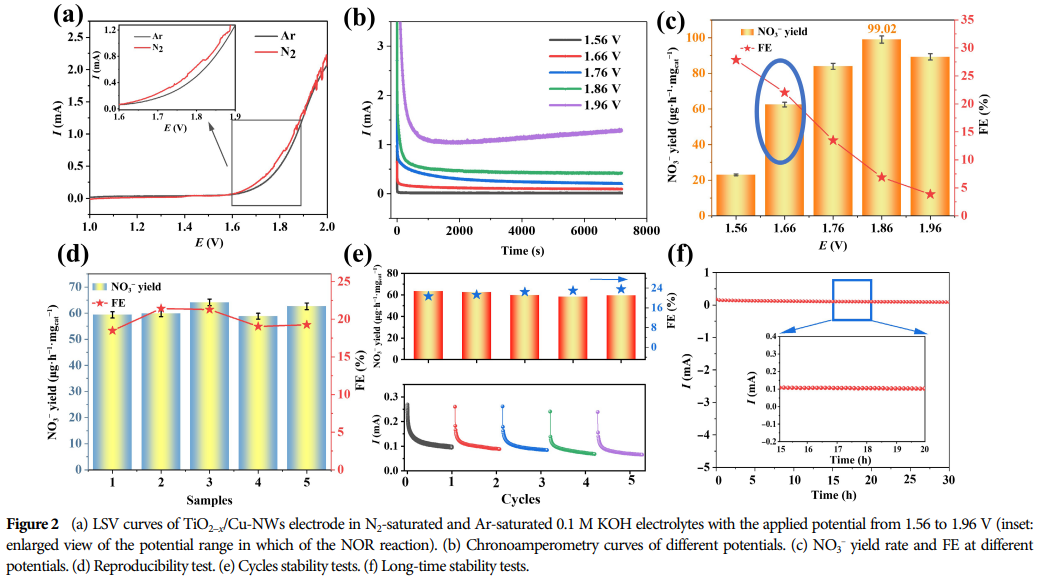
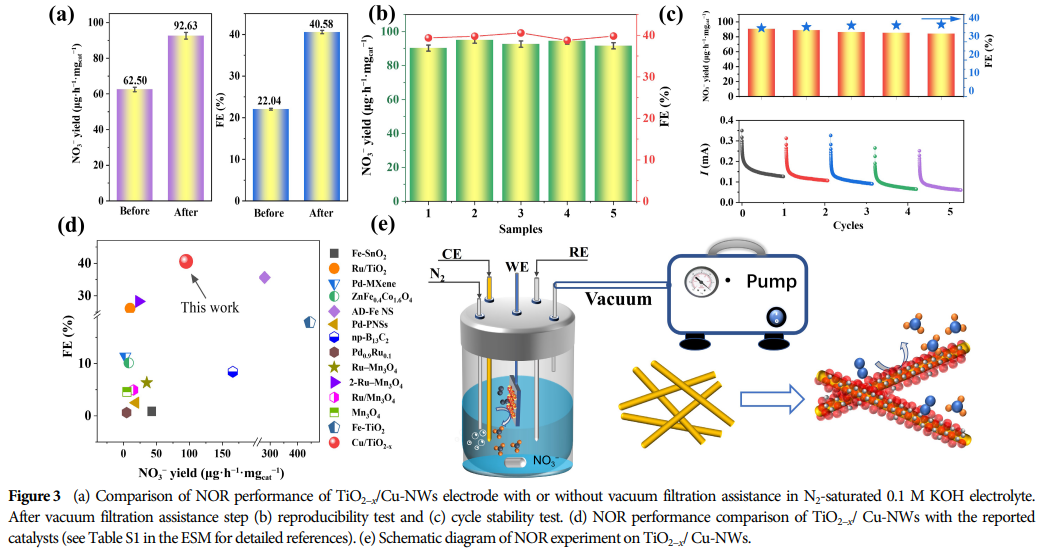
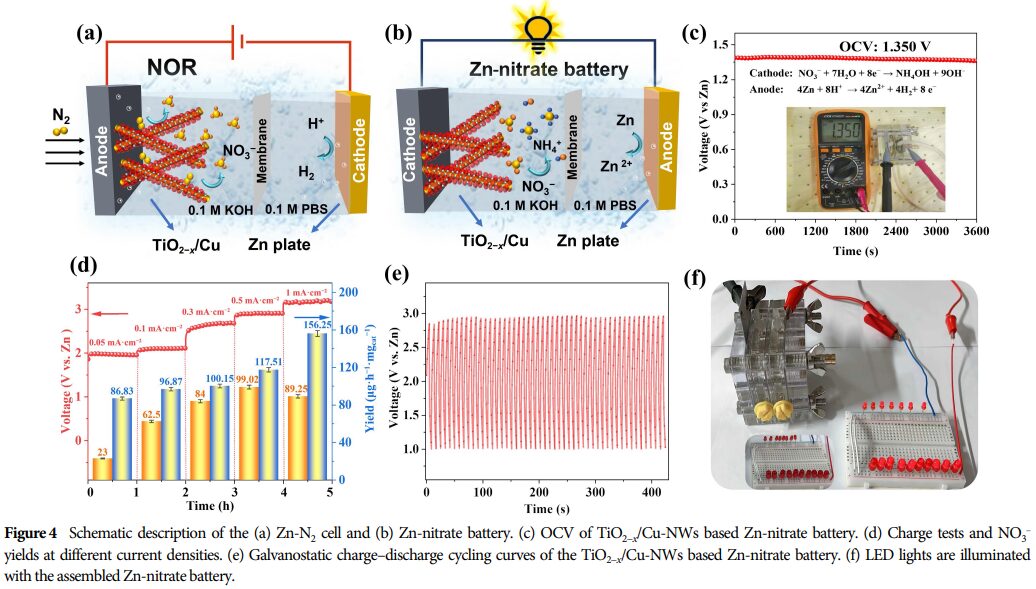
5. Research Significance
1. Green Synthesis of Nitrates
-
This study provides a new pathway for the green synthesis of nitrates, which has significant environmental friendliness and economic feasibility.
-
The excellent performance of the CuNWs@TiO2−x composite catalyst offers potential for industrial applications.
2. Advancement of Electrocatalytic Technology: This study promotes the development of the electrocatalytic nitrogen oxidation reaction field, providing new ideas and insights for the research of other electrocatalytic reactions.

6. Research Limitations
1. Catalyst Stability: Although the CuNWs@TiO2−x composite catalyst demonstrates excellent performance in the electrocatalytic nitrogen oxidation reaction, its long-term stability and regenerability require further study.
2. Optimization of Reaction Conditions: While vacuum-assisted conditions improve catalytic efficiency, they also increase experimental complexity and costs. Future exploration of milder and more efficient reaction conditions is necessary.
7. Future Research Directions
1. Catalyst Modification: Further modification of the CuNWs@TiO2−x composite catalyst is needed to enhance its catalytic activity and stability to meet industrial application demands.
2. Deepening Reaction Mechanism: In-depth exploration of the reaction mechanism and kinetics of the electrocatalytic nitrogen oxidation reaction will provide theoretical guidance for catalyst design and optimization.
This article is provided by the Hydrogen Energy Research Assistant.
Original link:
https://www.sciopen.com/article/10.1007/s12274-023-6126-8
▲ Programmable Dual-Module Membrane Electrode Spraying Machine