

1 In₂O₃ Electrocatalytic Performance Limitations
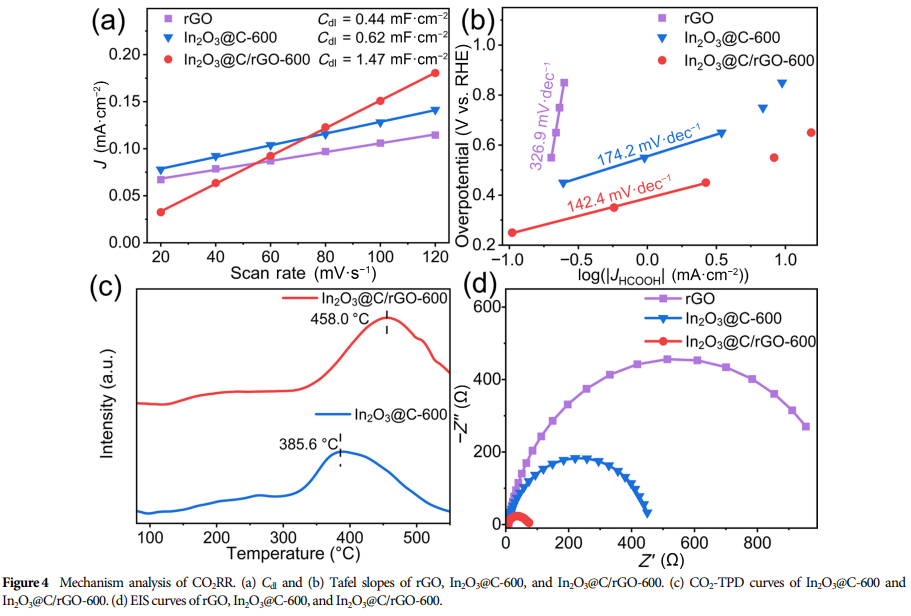
① Conductivity Issues: As an electrocatalyst, while In₂O₃ can effectively convert CO₂ to HCOOH, its poor conductivity limits charge transfer efficiency.
② Aggregation Issues: In₂O₃ particles are prone to aggregation during synthesis, reducing the exposure of active sites and further diminishing catalytic performance.
③ Research Goals: To address the conductivity and aggregation issues of In₂O₃ by constructing composites, thereby improving the Faradaic efficiency and energy efficiency of CO₂ reduction to HCOOH.
2 Composite Material Design
① Utilizing Graphene Oxide (GO) Template Effect: Achieving uniform anchoring of In₂O₃@C nanocubes on reduced graphene oxide (rGO) through the template effect of GO.
② InBDC Self-Sacrificial Template: Using InBDC as a self-sacrificial template to synthesize In₂O₃@C/rGO composites through pyrolysis.
3. Research Methods
-
Material Synthesis
① Synthesis of GO and InBDC: Detailed description of the synthesis steps for GO and InBDC.
② Preparation of In₂O₃@C/rGO Composites: Forming In₂O₃@C nanocubes on rGO through the pyrolysis of InBDC.
II. Characterization and Electrochemical Measurements
① Material Characterization: Using SEM, TEM, XRD, and other methods to characterize the morphology, structure, and composition of the materials.
② Electrochemical Measurements: Evaluating the electrocatalytic performance of the materials through linear sweep voltammetry (LSV), electrochemical impedance spectroscopy (EIS), and other methods.

3. Research Results
-
Structural Characterization:
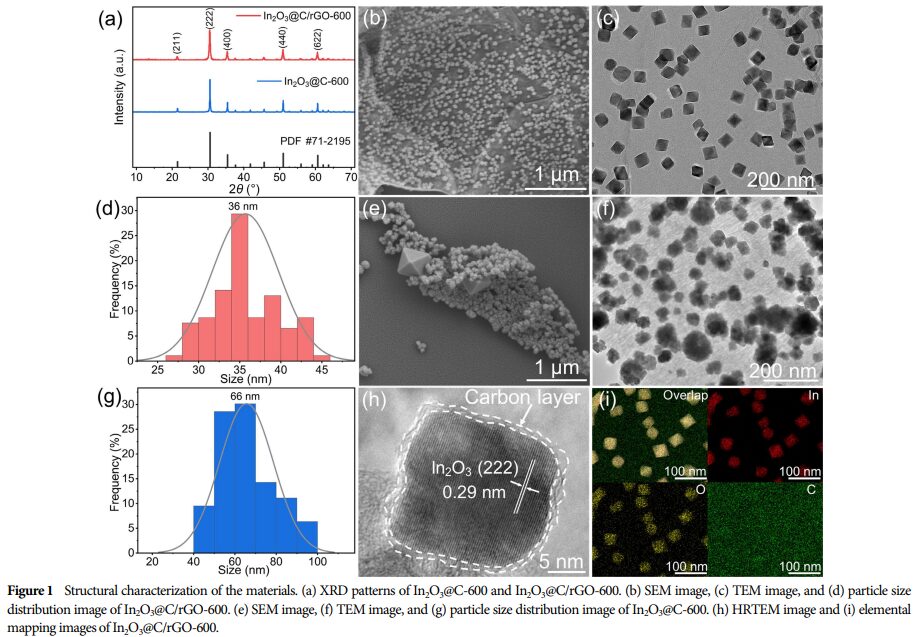
① Uniform Anchoring of In₂O₃@C Nanocubes: SEM and TEM images show that In₂O₃@C nanocubes are uniformly distributed on rGO, effectively preventing aggregation.
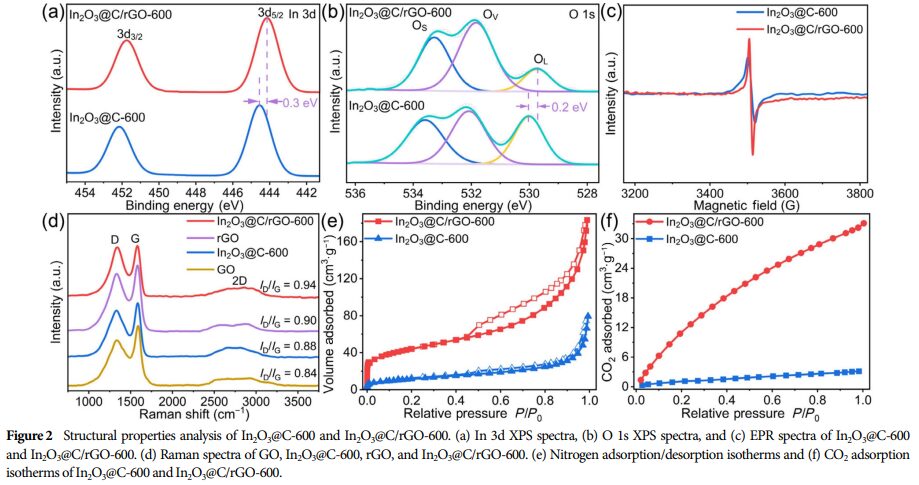
② High Specific Surface Area: The composite material has a high specific surface area, which is beneficial for exposing more active sites.
ii. Electrochemical Performance:
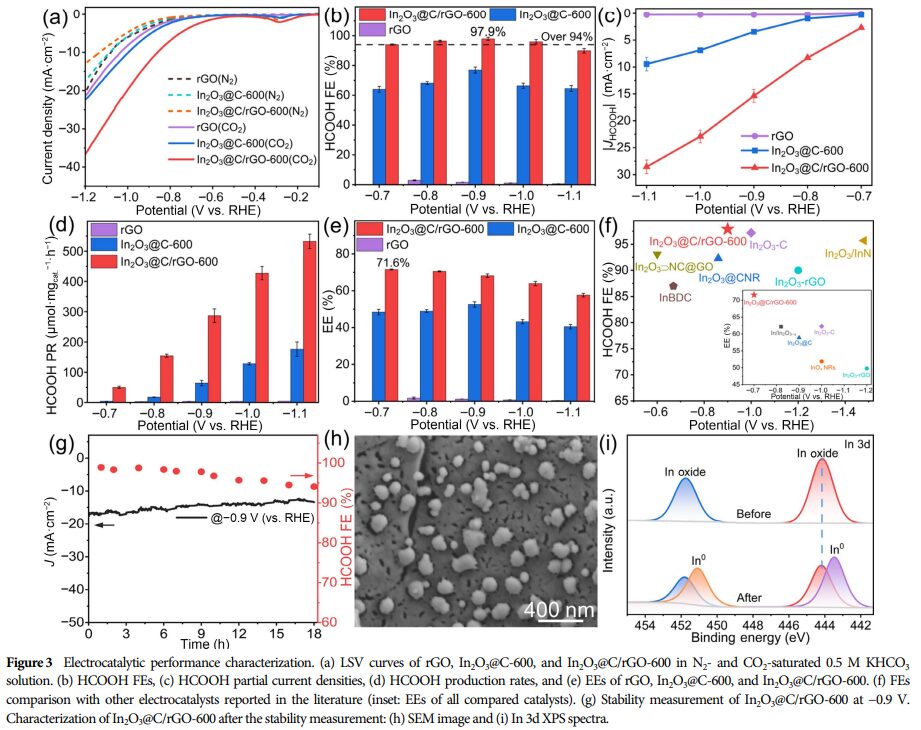
① High HCOOH Faradaic Efficiency: Within the potential range of -0.7 to -1.0 V (vs. RHE), the Faradaic efficiency for HCOOH exceeds 94.0%, reaching a maximum of 97.9% at -0.9 V (vs. RHE).
② Excellent Energy Efficiency: At -0.7 V (vs. RHE), the cathodic energy efficiency is 71.6%.
4. Visual Guide

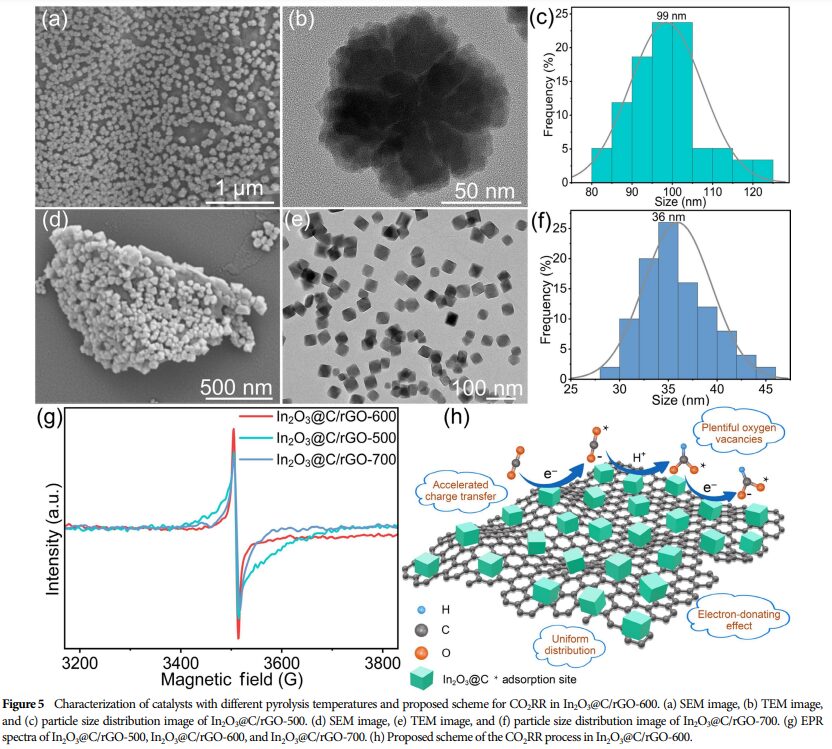
5. Research Significance
-
Improving CO₂ Reduction Efficiency: The In₂O₃@C/rGO composite material achieves efficient CO₂ reduction to HCOOH, providing new pathways for CO₂ utilization.
-
Advancing Electrocatalytic Technology Development: This research addresses the conductivity and aggregation issues of In₂O₃ through composite material construction, offering new insights for the design of electrocatalysts.
6. Research Limitations
1. Long-term Stability Not Addressed: Although the composite material exhibits excellent electrochemical performance, the article does not mention the long-term stability of the material, which is an important indicator for assessing the practicality of the catalyst.
2. Insufficient Discussion of Reaction Mechanism: The article lacks in-depth exploration of the reaction mechanism for CO₂ reduction to HCOOH, which requires further investigation in the future.
7. Future Research Directions
-
Optimizing Composite Material Structure: Further optimizing the electrocatalytic performance by adjusting the ratio and morphology of In₂O₃@C and rGO in the composite material.
-
Exploring More Application Scenarios: Applying In₂O₃@C/rGO composite materials in other electrocatalytic reactions, such as hydrogen evolution and nitrogen reduction.
-
In-depth Study of Reaction Mechanism: Using in-situ characterization and theoretical calculations to explore the reaction mechanism of CO₂ reduction to HCOOH, providing theoretical guidance for catalyst design.
In summary, this study constructs In₂O₃@C/rGO composite materials to achieve efficient electrocatalytic reduction of CO₂ to HCOOH, providing new insights for CO₂ utilization and the development of electrocatalytic technology. However, further exploration of the long-term stability, reaction mechanism, and additional application scenarios of the composite material is still needed.