Skip to content
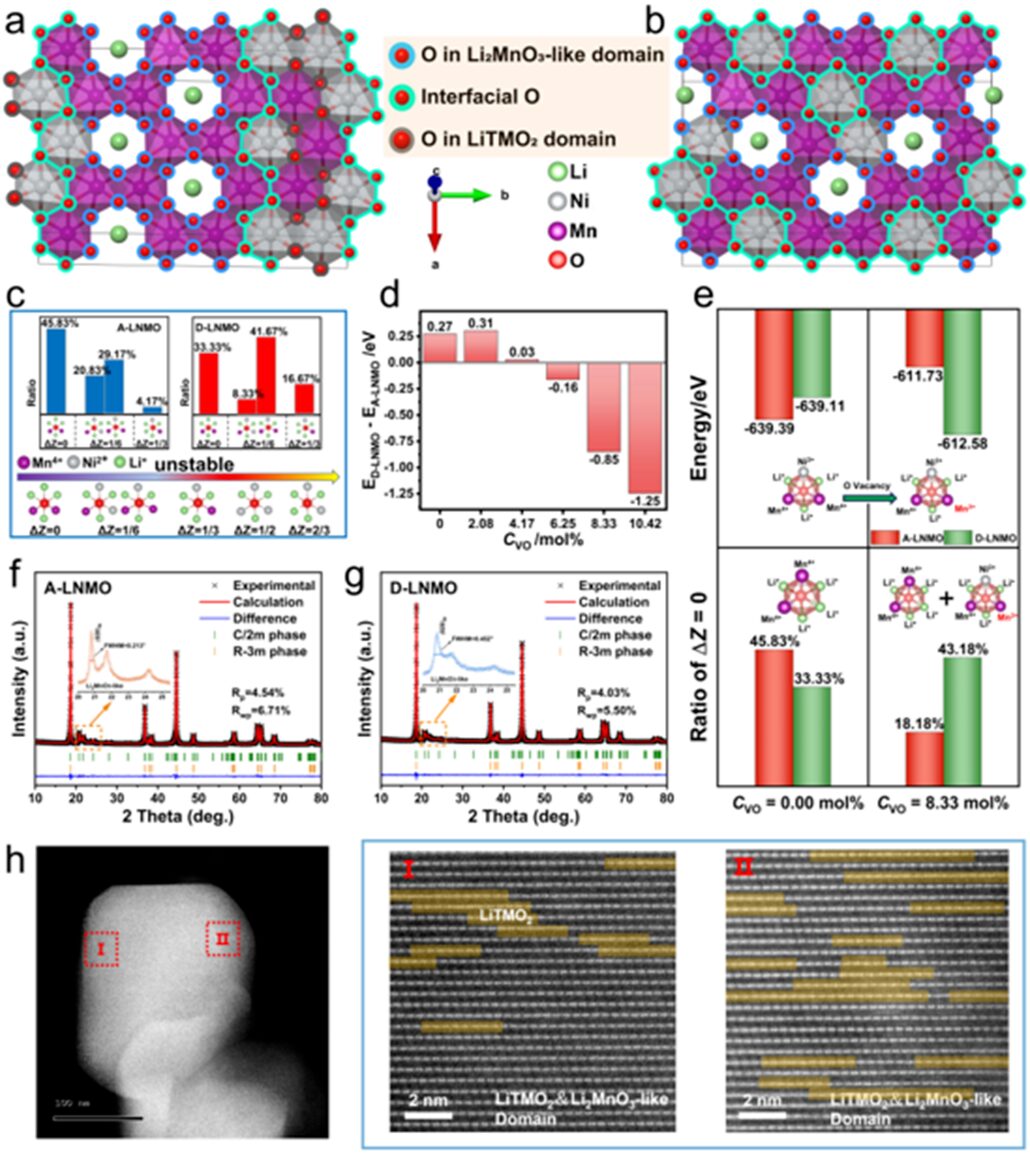
The key to controlling the oxygen oxidation-reduction chemistry is to adjust the chemical environment of the lattice oxygen with redox activity, namely the Li2MnO3-like domains.Recent studies have shown that aggregated large-sizeLi2MnO3-like domains can lead to irreversible TM migration and oxygen oxidation-reduction, resulting in notorious capacity fading and voltage decay. According to Pauling’s rules, the coordination configuration of OMn2Li4 with Mn4+ is stable, making the formation of aggregated Li2MnO3-like domains quite simple. Some studies indicate that aggregated Li2MnO3-like domains weaken the covalency and stability of the TM-O bond, leading to excessive oxygen oxidation and poor electrochemical stability. Therefore, controlling the distribution and size of Li2MnO3-like domains has become an effective method to enhance the electrochemical performance of LLOs. Related modification methods such as component adjustment, local structure design, and cobalt infiltration into Li2MnO3 domains have been employed to disrupt the aggregation of Li2MnO3-like domains. Although these strategies can effectively enhance the electrochemical performance of LLOs, their underlying mechanisms remain unclear.
Recently, a research group led by Professors An Shengli and Jia Guixiao from Inner Mongolia University of Science and Technology collaborated with Professor Qiu Xinqing’s group at Tsinghua University to report a stabilizing effect of weak σ–π–σ interactions on the oxidation-reduction of oxygen in lithium-rich cathode materials. This work synthesized Li2MnO3-like domains with good dispersion (D-LNMO) and aggregated Li2MnO3-like domains (A-LNMO) in LNMO materials by controlling the oxygen partial pressure during the high-temperature calcination process. Based on density functional theory (DFT) calculations and Pauling’s rules, we demonstrated that low oxygen partial pressure can disperse Li2MnO3-like domains by forming stable ONiMn4+Mn3+Li3 coordination configurations. Additionally, the mechanism of oxygen oxidation-reduction was revealed through the orbital interactions between two O2p at the atomic level. The weak σ–π–σ interaction triggered a new mechanism for oxygen oxidation-reduction in the OTM2MnLi3 (TM = Ni and Mn) coordination configuration. Due to the abundance of interfacial lattice oxygen in D-LNMO, more lattice oxygen participates in charge compensation, thus suppressing the release of lattice oxygen and irreversible structural transformation. Therefore, the electrochemical performance of D-LNMO is significantly superior to that of A-LNMO. This work provides valuable insights for developing high-performance lithium-rich cathodes. The related results were published in the international journal Nano Energy under the title “Weak σ–π–σ Interaction Stabilizes Oxygen Redox by Activating Interfacial Oxygen towards High-Performance Li–Rich Layered Oxide Cathodes.” This article is co-first authored by Guo Xin, a master’s student from the School of Materials and Metallurgy at Inner Mongolia University of Science and Technology, and Li Jie, a doctoral student from the Key Laboratory of Organic Optoelectronics and Molecular Engineering at Tsinghua University.
It is widely believed that Pauling’s electrostatic valence rule proves the stability of anionic coordination environments in stable ionic crystals. A semi-quantitative equation for thermodynamic assessment of structural stability is provided:
Where ZA and ZC are the valences of anions and cations, respectively, and n is the coordination number of cations. The more structures with ∆Z=0, the more stable they are.
From this formula, A–LNMO is more stable than D–LNMO. This is because the valences of Li, Ni, Mn, and O in LNMO are +1, +2, +4, and -2, respectively, resulting in more O-centered coordination configurations when ΔZ=0 for A-LNMO. However, when oxygen vacancies are introduced, the valence state of Mn changes from +4 to +3. When CVO=8.3mol%, a large number of ΔZ=1/6 ONiMn24+Li3 configurations transform into the stable configuration of ΔZ=0 ONiMn4+Mn3+Li3, thereby stabilizing D-LNMO. This indicates that synthesizing D-LNMO with well-dispersed Li2MnO3-like domains can be achieved by introducing a higher concentration of oxygen vacancies.
Figure 1 shows that according to Pauling’s rules, it is found that introducing oxygen vacancies leads to greater stability in D-LNMO.
To verify this, we synthesized D–LNMO by lowering the oxygen partial pressure during high-temperature calcination. For comparison, A-LNMO was obtained by calcining in air. The chemical composition tested by ICP-OES was very consistent with the theoretical values. Through XRD patterns and Rietveld refinement results of the two-phase model, as well as HAADF-STEM, we comprehensively analyzed and observed that D-LNMO has more dispersed and smaller Li2MnO3-like domains.
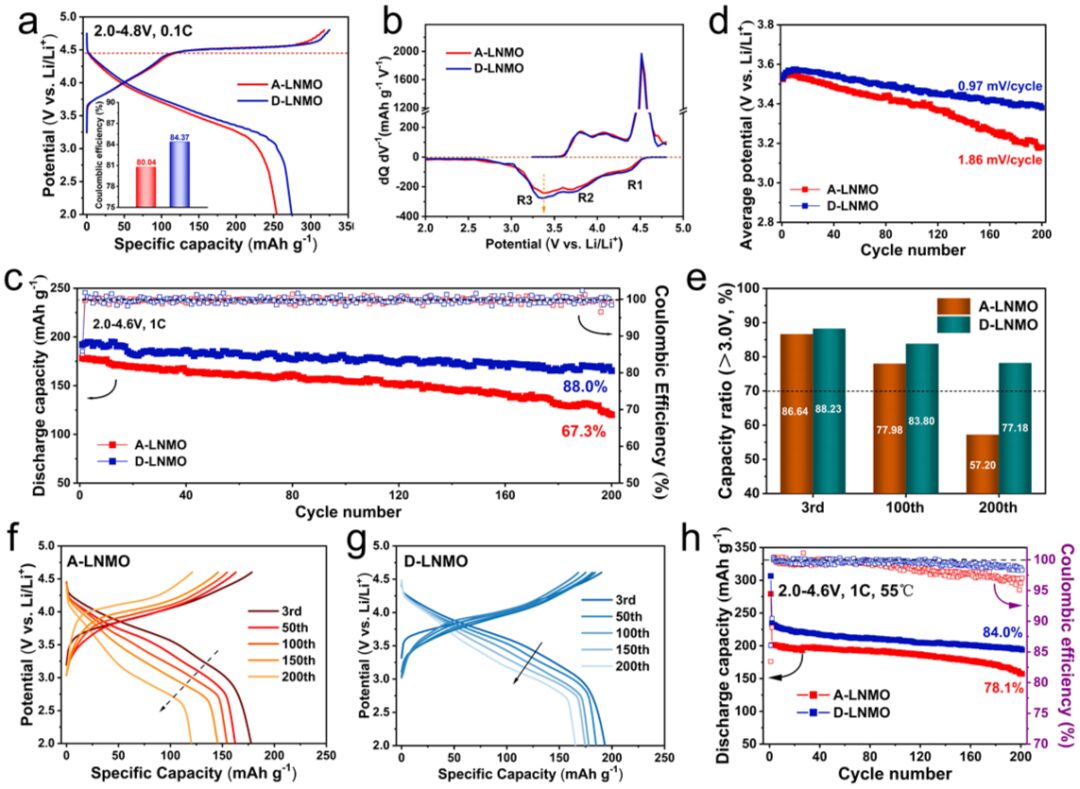
By experimentally testing the electrochemical performance of both, it was found that D-LNMO with dispersed small Li2MnO3-like domains exhibits excellent electrochemical performance, better thermal stability, and its excellent cycling performance also proves its highly reversible oxygen oxidation-reduction and stable structure.
Figure 2 compares the electrochemical performance of A-LNMO and D-LNMO.
Next, to find the source of this reversible oxygen oxidation-reduction and structural stability, DFT calculations were used to analyze the distribution and direction of charge. Through local charge density maps, it was found that D-LNMO has more oxygen participating in oxidation compared to A-LNMO, and these oxygens are distinct from those in the traditional Li-O-Li configuration. They exist between Li2MnO3-like and LiTMO2 domains, which we refer to as interfacial oxygen. By observing the charge density direction of interfacial oxygen and the changes in bond lengths between oxygen, we analyzed and proposed the weak σ–π–σ interaction at the two-domain interface, that is, through σ interactions between the oxygens in the two Li-O-Li configurations and π interactions between the two interfacial oxygens, this interaction activates the interfacial oxygen to participate in oxidation. Especially in D–LNMO, the dispersed Li2MnO3-like domain structure leads to a large number of weak σ–π–σ interactions, while such interactions are limited to a few interfacial oxygens in A–LNMO.
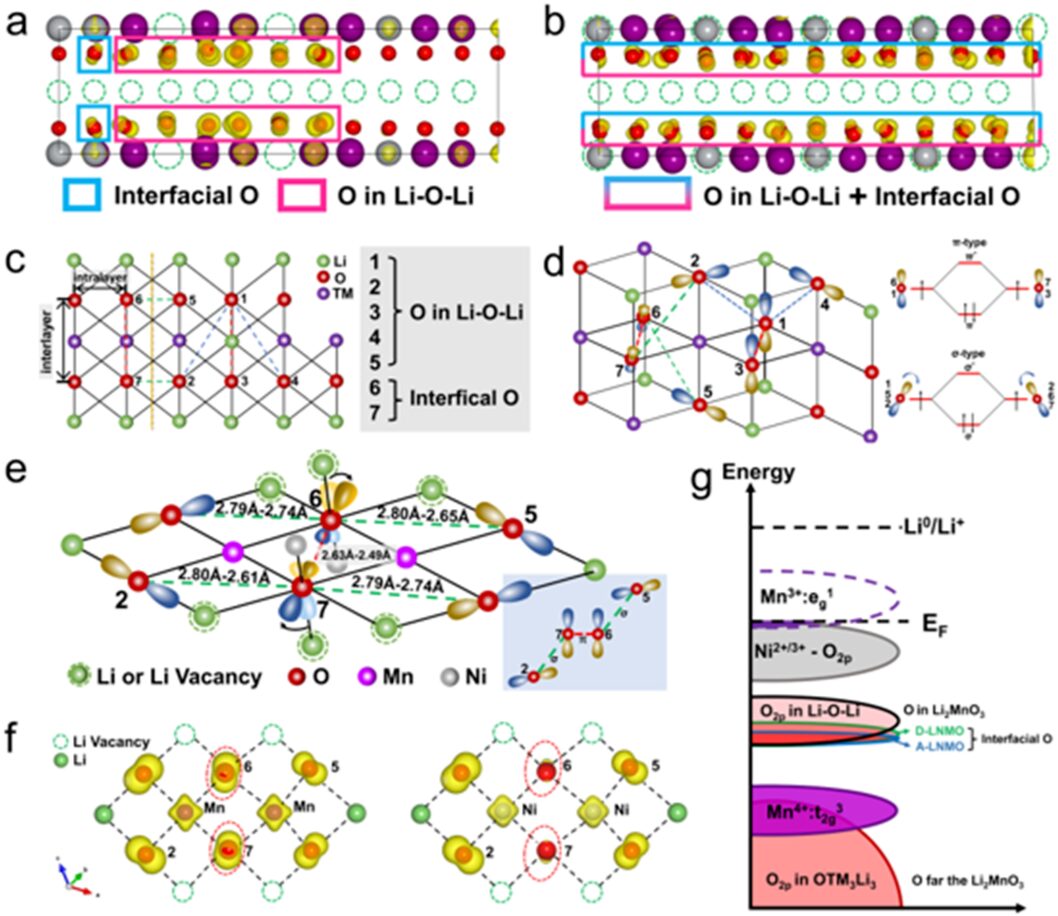 Figure 3 illustrates the formation mechanism of the weak σ–π–σ interaction.
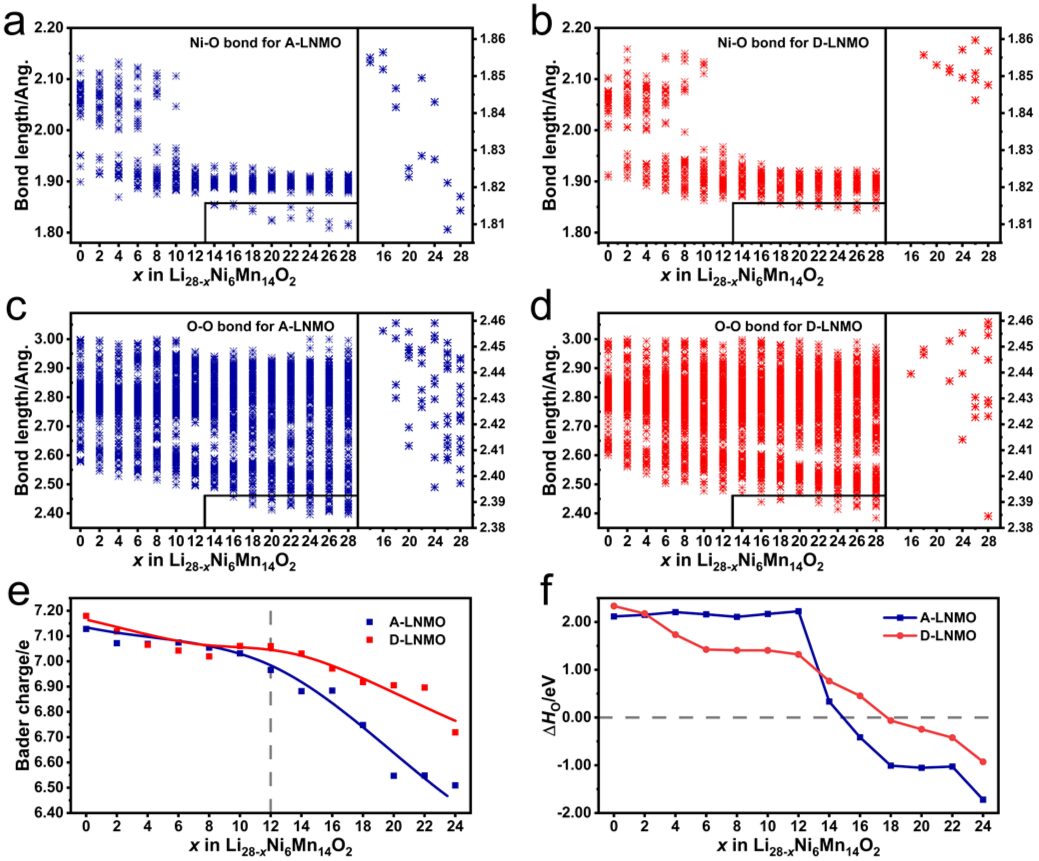
Finally, a series of DFT calculations were performed, showing bond length distribution, Bader charge, oxygen vacancy formation enthalpy, TM migration, etc., all pointing to the superior performance of D-LNMO.
Figure 4 shows the bond length distribution, Bader charge, and oxygen vacancy formation enthalpy.
D-LNMO’s Ni-O and O-O short bond length distributions are fewer, Bader charge variations are smaller, and the oxygen vacancy formation enthalpy becomes negative later, all of which prove that D-LNMO alleviates the oxidative load on oxygen ions, suppressing the release of lattice oxygen.
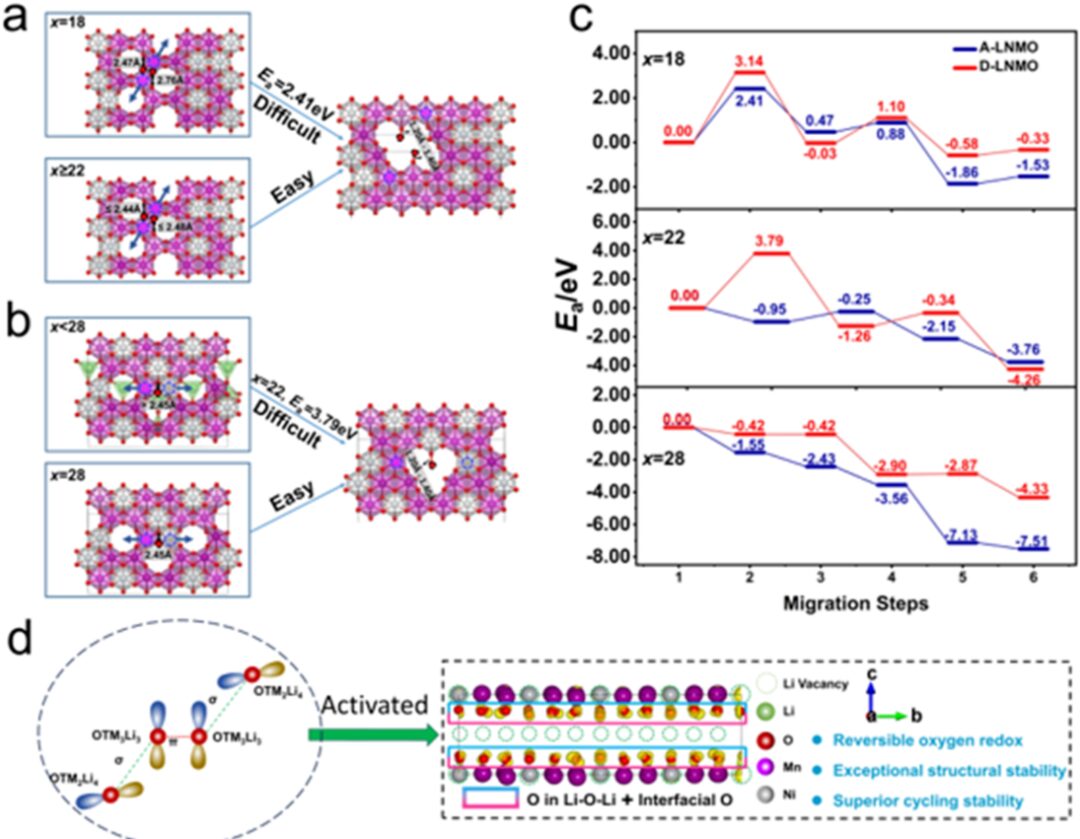
Figure 5 illustrates the migration principle and energy barrier of TM and the schematic diagram of this principle.
Finally, to kinetically explain the structural stability of D-LNMO, we used the CI-NEB method to find the transition state and calculate it, discovering that the TM migration energy barrier of D-LNMO is higher, which also proves that D-LNMO has better structural stability than A-LNMO. It is proposed that the oxidation of oxygen may promote the migration of TM.
This work reveals a new mechanism of oxygen oxidation, namely the oxygen oxidation-reduction triggered by weak σ–π–σ interactions in the OTM2MnLi3 (TM=Ni, Mn) configuration. Specifically, the lattice oxygen at the interface of Li2MnO3-like domains and LiTMO2 domains can be activated. Therefore, the well-dispersed and smaller Li2MnO3-like domains in D-LNMO can trigger more lattice oxygen to participate in charge compensation, thereby alleviating the oxidative load on oxygen ions, suppressing the release of lattice oxygen, and delaying irreversible structural transformation. Benefiting from this, D-LNMO provides a high discharge capacity of 274.3 mAh g-1 at 0.1C, maintaining up to 88.0% capacity retention after 200 cycles at 1C, with a small voltage decay of 0.97 mV per cycle. This work establishes the relationship between the distribution of Li2MnO3-like domains and the stability of oxygen oxidation-reduction, guiding the synthesis of high-performance LLOs.
Guo, X.; Li, J.; Zhang, Y.; Zhang, X.; Liu, J.; Li, W.; Lu, L.; Jia, G.; An, S.; Qiu, X. Weak σ–π–σ Interaction Stabilizes Oxygen Redox towards High-Performance Li–Rich Layered Oxide Cathodes. Nano Energy2024, DOI:10.1016/j.nanoen.2024.109390
Figure 3 illustrates the formation mechanism of the weak σ–π–σ interaction.
Finally, a series of DFT calculations were performed, showing bond length distribution, Bader charge, oxygen vacancy formation enthalpy, TM migration, etc., all pointing to the superior performance of D-LNMO.
Figure 4 shows the bond length distribution, Bader charge, and oxygen vacancy formation enthalpy.
D-LNMO’s Ni-O and O-O short bond length distributions are fewer, Bader charge variations are smaller, and the oxygen vacancy formation enthalpy becomes negative later, all of which prove that D-LNMO alleviates the oxidative load on oxygen ions, suppressing the release of lattice oxygen.
Figure 5 illustrates the migration principle and energy barrier of TM and the schematic diagram of this principle.
Finally, to kinetically explain the structural stability of D-LNMO, we used the CI-NEB method to find the transition state and calculate it, discovering that the TM migration energy barrier of D-LNMO is higher, which also proves that D-LNMO has better structural stability than A-LNMO. It is proposed that the oxidation of oxygen may promote the migration of TM.
This work reveals a new mechanism of oxygen oxidation, namely the oxygen oxidation-reduction triggered by weak σ–π–σ interactions in the OTM2MnLi3 (TM=Ni, Mn) configuration. Specifically, the lattice oxygen at the interface of Li2MnO3-like domains and LiTMO2 domains can be activated. Therefore, the well-dispersed and smaller Li2MnO3-like domains in D-LNMO can trigger more lattice oxygen to participate in charge compensation, thereby alleviating the oxidative load on oxygen ions, suppressing the release of lattice oxygen, and delaying irreversible structural transformation. Benefiting from this, D-LNMO provides a high discharge capacity of 274.3 mAh g-1 at 0.1C, maintaining up to 88.0% capacity retention after 200 cycles at 1C, with a small voltage decay of 0.97 mV per cycle. This work establishes the relationship between the distribution of Li2MnO3-like domains and the stability of oxygen oxidation-reduction, guiding the synthesis of high-performance LLOs.
Guo, X.; Li, J.; Zhang, Y.; Zhang, X.; Liu, J.; Li, W.; Lu, L.; Jia, G.; An, S.; Qiu, X. Weak σ–π–σ Interaction Stabilizes Oxygen Redox towards High-Performance Li–Rich Layered Oxide Cathodes. Nano Energy2024, DOI:10.1016/j.nanoen.2024.109390