Background Introduction
Results Overview
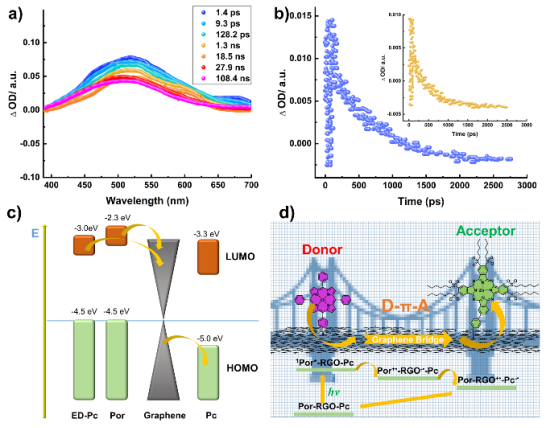
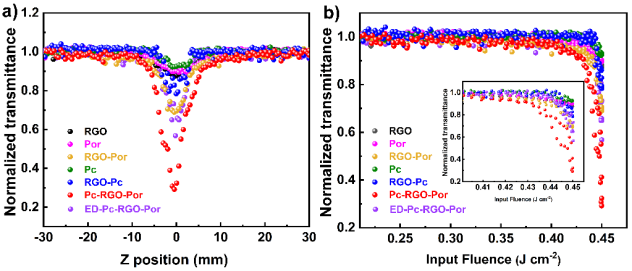
This article reports a D-π-A type porphyrin-phthalocyanine covalently bifunctionalized graphene nonlinear nano-conjugate material. Phthalocyanine with strong electron-withdrawing groups and tetra-phenyl porphyrin were sequentially linked to the surface of graphene through radical addition reactions. In the formed ternary hybrid system, graphene acts as a bridging agent, extending the charge-separated state of the hybrid system and enhancing the third-order nonlinear properties of this organic-inorganic nano-conjugate material. In this novel structure, six strong electron-withdrawing sulfonyl groups substituted asymmetric amino phthalocyanine (Pc) act as electron acceptors, while 5,10,15,20-tetraphenylporphyrin (Por) plays the role of electron donor, and reduced graphene oxide (RGO) uniquely serves as an electron bridge for the first time. To date, the simultaneous introduction of electron-donating porphyrins and electron-accepting phthalocyanines on the surface of graphene in a ternary system has not been explored. Transient absorption spectroscopy indicates that in Por-RGO-Pc, the D-π-A gradient structure leads to cascading charge transfer, extending the lifetime of charge separation and improving charge transfer efficiency, consistent with the highly quenched (99.5%) fluorescence results. Z-scan results show that the significant nonlinear absorption of the ternary system is markedly enhanced, with a nonlinear absorption coefficient reaching up to 8.7ⅹ102 cm/GW on the nanosecond timescale. In this work, the design strategy of the D-π-A ternary system provides a new example for the next generation of graphene-based optoelectronic devices, offering significant insights for designing better-performing optical biomimetic devices, solar cells, photocatalysis, and particularly optical limiting devices.
Illustrated Guide
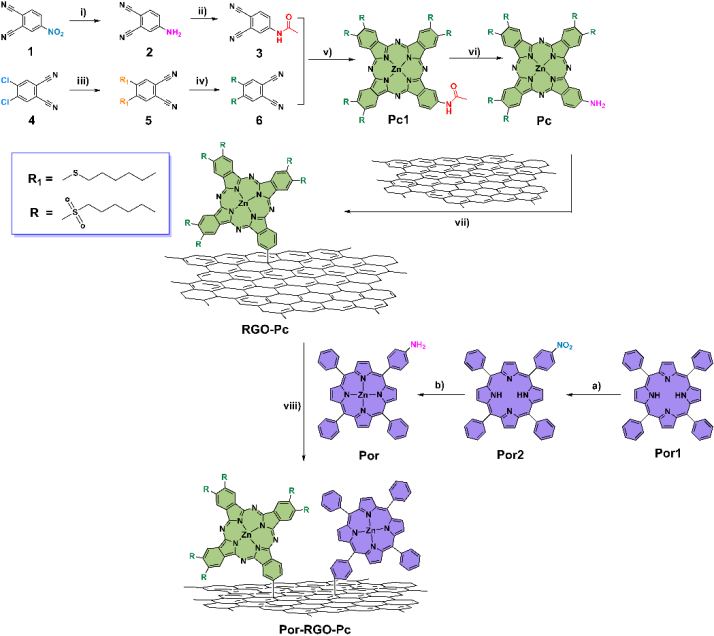
Figure 1. Schematic diagram of the synthesis of D-π-A type porphyrin-phthalocyanine bifunctionalized graphene nano-conjugate material Por-RGO-Pc.
Author Introduction
Professor Zhang Chi is currently the Dean of the School of Chemical Science and Engineering at Tongji University, Director of the China-Australia International Joint Research Center for Functional Molecular Materials, Director of the National International Joint Research Center for Light Responsive Functional Materials under the Ministry of Science and Technology, Member of the Materials Division of the Science and Technology Committee of the Ministry of Education, Member of the Expert Committee of the China Overseas Chinese Federation, and Fellow of RSC, IMMM, IET, and RACI. Professor Zhang Chi’s research group has long been engaged in the nonlinear optical and optoelectronic functional research of functional materials, proposing new methods for optimizing functional cluster frameworks and enhancing material optical limiting performance through secondary molecular modification, revealing the modulation rules of functional organic conjugated units on the optical nonlinearity of cluster materials, and developing theories on optical saturation/reverse saturation absorption that integrate conjugated unit modification and modulation of the optical activity of two-dimensional carbon-based materials. He has been evaluated as representing the highest research level in the field of optical functional materials spectroscopy and diffraction method experimental research, and the organic-inorganic conjugated materials created have great potential to become photonic or optoelectronic devices, pushing forward the academic frontier at the nanoscale. To date, he has published over 330 papers in internationally renowned academic journals, with over 8000 citations and 32 authorized invention patents. He leads the Ministry of Education’s innovation team and the key innovation team in key areas of the Ministry of Science and Technology, and has presided over over 20 national and international research projects, including the National Natural Science Foundation’s outstanding youth, key, and general projects, key international cooperation projects from the Ministry of Science and Technology, and ARC Discovery Project, DIISR Intl. Sci. Linkages.
Article Information
Fu L, Li H, Fang Y, et al. Cascading electron transfer and photophysics in a donor-π-acceptor graphene nanoconjugate. Nano Research, 2022, https://doi.org/10.1007/s12274-022-5167-8.

Scan the QR code or click the bottom left “Read the original text” to access the full text.
Follow us on Bilibili, Video accounts, and official website for more exciting content!

Bilibili

Video Account

Official Website