In cancer treatment, there are three targeted approaches to enhance the specificity and anti-tumor activity of therapies. Targeted formulations can be designed to inhibit proteins expressed by tumor cells, such as receptors or enzymes; another approach involves binding effector molecules (e.g., ADCs, bispecific antibodies, or CAR-T) to molecules overexpressed on the surface of tumor cells, synergistically inhibiting tumor cell division while providing cytotoxic payloads or stimulating tumor-directed immune responses; the third method uses Peptide-Drug Conjugates (PDCs) to drive the accumulation of toxic payloads within tumor cells.

Antibody-drug conjugates (ADCs) have become a hot research topic in the field of tumor treatment in recent years. As more companies enter the ADC drug field, there has been a phenomenon of concentration on targets and indications. At the same time, challenges such as the toxic side effects of ADC drugs, high production costs, and suboptimal efficacy against certain solid tumors have gradually emerged. PDCs utilize peptides as tumor-targeting carriers, and compared to ADCs, they are easier to synthesize, allow for straightforward structural modifications, and support rational drug design to improve bioavailability, affinity, and stability. Additionally, peptides have lower immunogenicity. Therefore, Peptide-Drug Conjugates (PDCs) represent a new generation of targeted therapeutic drugs following ADCs.

Source: Yaozhi News
Structure and Mechanism of Action of PDCs
Peptide-drug conjugates (PDCs) are drugs that covalently link drugs to peptide sequences through special linkers, consisting of three parts: homing peptides, linkers, and cytotoxic payloads.

 Cytotoxic Payload
Cytotoxic Payload
After PDCs enter cells, the toxic drug is the primary agent that ultimately leads to the death of target cells; therefore, the toxicity and physicochemical properties of the toxic drug can directly affect the drug’s ability to kill tumors, thus influencing efficacy. The cytotoxic agent used in conjugation must meet the following four requirements: a clear mechanism of action, small molecular weight, high cytotoxicity, and retention of anti-tumor activity after chemical conjugation with the peptide.
The effective payload of PDCs can take various forms, with the most common being cytotoxic drugs such as paclitaxel and gemcitabine; radioactive nuclides are used for precise diagnosis of diseases and targeted radiotherapy of tumors. For instance, diagnostic radioactive isotopes include fluorine-18 (18F), copper-64 (64Cu), gallium-68 (68Ga), and iodine-123 (123I), while therapeutic radioactive nuclides include indium-111 (111In), yttrium-90 (90Y), and lutetium-177 (177Lu). PDCs serve as targeted radiotherapy drugs, and in addition, proteins, antibodies, peptides, nucleic acids, and PROTACs can also be conjugated with targeting peptides.
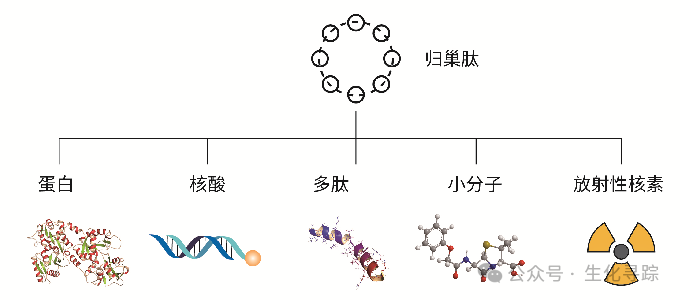
Types of PDC Payloads
Homing Peptides for Tumors
Homing peptides are specifically targeted to overexpressed protein receptors in tumor tissues. They directly deliver the drug to target cells, limiting off-target delivery of chemotherapeutic agents. The peptides used in PDCs can be divided into two categories: cell-penetrating peptides (CPPs) and cell-targeting peptides (CTPs). The distinction lies in that the former can transport drugs across cell membranes, while the latter can specifically bind to receptors on target cells. The ideal peptide for PDCs should have strong target binding affinity, high stability, low immunogenicity, efficient internalization, and a long plasma half-life. Appropriate methods are also needed to assess the relevant properties of the peptides.
Cell-Penetrating Peptides (CPPs): Short peptides containing 5 to 40 amino acids that enter cells through endocytosis without disrupting membrane integrity, exhibiting both positive charge and amphiphilicity in physiological environments, and have been widely used in drug delivery systems. Examples include Pep-1, Pentratin, PepFact14, and Transportan. However, their drawbacks include a lack of cell specificity, short action time, poor in vivo stability, and compatibility issues.
Cell-Targeting Peptides (CTPs): After binding to receptors on the surface of tumor cells, CTPs deliver PDC drugs into cells through receptor-mediated endocytosis. In lysosomes, the linker is cleaved by relevant enzymes or under specific chemical conditions (such as the low pH of the tumor microenvironment), releasing the naked drug to exert its tumor-killing effect. CTPs have high affinity for receptors overexpressed on tumor cell surfaces, and therapeutic drugs conjugated with CTPs can be transported and enriched at the target site, significantly reducing side effects. They can be classified into passive and active targeting; active targeting involves the specific delivery of drugs to diseased tissues by recognizing receptors or proteins specifically expressed in target tissues, while passive targeting refers to the passive accumulation of drugs in diseased tissues due to the properties of the drug delivery system itself or the characteristics of the target tissue. Most PDCs based on CTPs are delivered through active targeting mechanisms. Examples include PEGA, growth hormone-releasing hormone analogs, frog skin peptide analogs, and RGD peptides. Their drawbacks include susceptibility to degradation in vivo and poor cell penetration ability.
After selecting the peptide, due to the ease of degradation by enzymes or the intracellular environment, it is necessary to enhance its stability and cell permeability. Currently, commonly used methods include cyclic peptides, stapled peptides, or the introduction of non-natural amino acids to improve membrane permeability, chemical stability, and enzymatic hydrolysis stability, thereby reducing renal clearance rates for better absorption and utilization by patients. Additionally, formulation factors can be altered, such as using pH-sensitive acid-stable coatings to encapsulate orally administered peptide formulations to protect them from degradation by gastric acid and pepsin.

Linkers
The linker is the bridge connecting the tumor-homing peptide and the toxic molecule, determining the circulation time and stability of the PDC in the body. On one hand, it is necessary to ensure the integrity of the PDC during circulation before it reaches the tumor cells to prevent premature and non-specific release that could lead to off-target toxicity; on the other hand, once the PDC enters the target cell, the linker must ensure the effective release of the toxic drug to exert its killing effect. Depending on the drug release mechanism and the cleavage behavior of the linker, linkers can be mainly divided into non-cleavable linkers and cleavable linkers. The latter can be further divided into pH-sensitive, enzyme-sensitive, and redox-sensitive types based on their cleavage mechanisms, with enzyme-sensitive ester or amide bonds being widely used due to their selective cleavage in the tumor microenvironment or lysosomes.
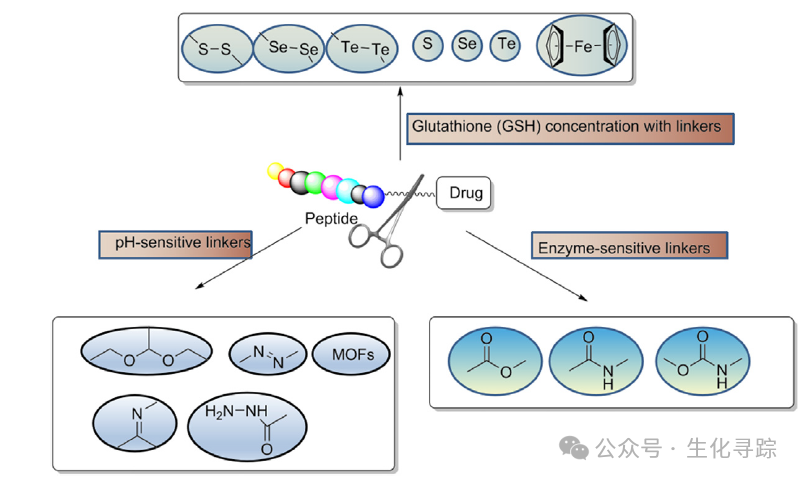
Mechanism of Action
The mechanism of action of PDCs is as follows: first, the homing peptide specifically targets and binds to overexpressed protein receptors on tumor cells, delivering the cytotoxic agent into the tumor cells through receptor-mediated internalization. Then, the linker is cleaved by enzymes or under specific chemical conditions (such as the low pH of the tumor microenvironment) inside the cell or in the tumor environment, releasing the naked drug to fully exert its tumor-killing effect.
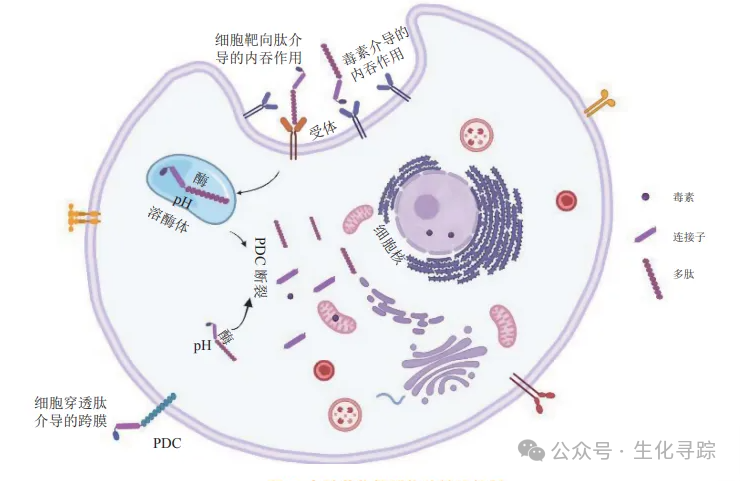
Current Research Status
Currently, only two PDCs have been approved by the FDA for clinical cancer treatment: Melflufen and 177Lu-dotatate. There are approximately 20 PDC drug pipelines entering clinical trial stages globally, with most indications related to cancer and the effective payloads primarily being MMAE, and most linkers being Amide, all of which are still in active development. The drugs with the fastest clinical progress are Paclitaxel trevatide (ANG-1005) and Zelenectide pevedotin (BT8009), both in Phase 3 clinical trials.

As an emerging class of conjugated drugs, there are currently two PDC drugs approved, with multiple pipelines advancing to late clinical stages, making it a new hotspot for global conjugated drug research and development. Compared to ADCs, PDCs have unique advantages in molecular weight, pharmacokinetics, payload drug selection, and production costs and cycles, making them promising candidates for the next generation of tumor-targeted therapeutic drugs following ADCs. Currently, PDCs still face limitations in stability and half-life, restricting their clinical use, and enhancing stability and prolonging half-life through cyclization, nanoparticles, and other technologies is a major direction for innovative PDC development.
Disclaimer: This article is based on publicly available information and is intended for knowledge sharing and dissemination, not constituting advice to any individual. If there are any infringements, please contact this public account for deletion.
For more exciting content, please follow and share this public account.