
In 2019, a CT imaging comparison from Gustave Roussy Hospital in Paris, France, caused a stir in the oncology community: a late-stage non-small cell lung cancer patient, who had failed both chemotherapy and immunotherapy, showed significant reduction in systemic metastatic lesions just two months after being enrolled in the clinical trial of Trastuzumab Deruxtecan (T-DXd), with intracranial lesions achieving marked radiological relief, leading to recommendations for long-term follow-up. This image opened up new possibilities for precise treatment of lung cancer for clinicians.
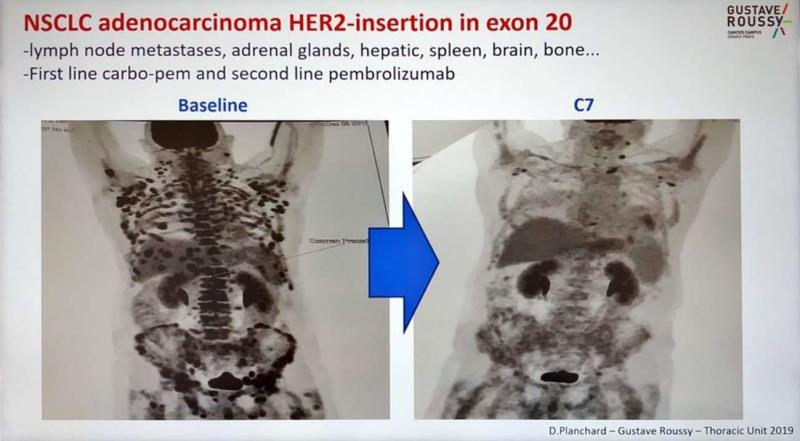
Case source: https://twitter.com/dplanchard/status/1266701523809165313
Three Keywords to Quickly Understand ADC Drugs
The full name of ADC is Antibody-Drug Conjugate, a class of targeted biopharmaceuticals that combine antibodies and cytotoxic drugs (payloads) through linkers. Each component of ADC serves a different function:1-2
Precise Targeting: Antibody part is the main framework and navigation system of the ADC drug, responsible for selectively delivering the ADC molecule to the surface of tumor cells and entering the cells through receptor-mediated endocytosis.
Safe Delivery: Linker is responsible for efficiently releasing toxic small molecules either inside or on the surface of tumor cells, with its chemical properties determining how and when the toxic small molecules are released. The stability of the linker significantly affects the safety and efficacy of ADC drugs, determining whether the toxic small molecules are delivered inside tumor cells or may be prematurely released during transport and distributed to other tissues via plasma, causing toxicity.
Efficient Killing: Cytotoxic Drug is the warhead that exerts cytotoxic effects and destroys cancer cells after being internalized into the cancer cells, achieving efficient clearance of cancer cells.
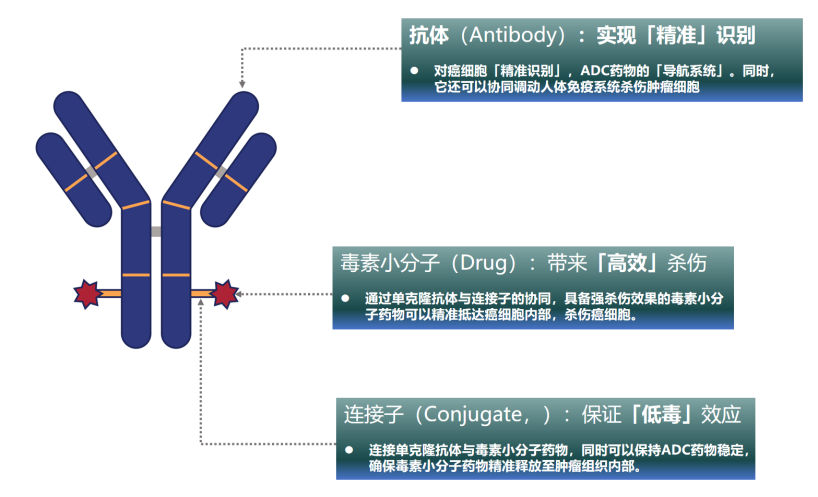
Diagram of ADC Drug Structure
Precise Targeting of Lung Cancer, The Therapeutic Revolution of ADC Drugs
Although systemic chemotherapy, targeted therapy, and immunotherapy can treat advanced lung cancer, many patients ultimately experience disease progression. Now, this emerging targeted therapy method, ADC, delivers cytotoxic drugs directly to cancer cells, minimizing off-target effects, providing a promising new treatment option for lung cancer (Tawfiq RK 2025)2.
The therapeutic advantages of ADC drugs are mainly reflected in:3
-
Single-target specificity of ADC drugs significantly reduces off-target toxicity and greatly expands the therapeutic window;
-
High local concentration release of cytotoxins in tumors enhances killing efficacy while avoiding side effects from systemic exposure;
-
When certain ADC drugs release payloads that are permeable or transmembrane, they may also induce a bystander effect*, further enhancing the efficacy of ADCs. Additionally, the bystander effect of these drugs may alter the tumor microenvironment, further enhancing the killing effect of ADCs.
*Bystander Effect: The bystander effect of ADC refers to the ability of certain ADC drugs to spread the payload from antigen-positive tumor cells to adjacent antigen-negative tumor cells, thereby enhancing antitumor activity. This effect allows ADCs to function even in tumors with uneven target antigen expression, as the payload can diffuse from antigen-positive cells that have bound the ADC to adjacent antigen-negative cells, killing them in the process.4.
Successful development of ADC drugs relies on selecting the right antigen targets and designing precise antibodies. Ideal targets are those that are highly expressed on cancer cells but have low expression levels in normal cells. Currently, popular targets for non-small cell lung cancer (NSCLC) mainly include HER2, TROP2, c-MET, and EGFR, with HER2’s low internalization and degradation rates making it a primary ADC target for NSCLC (Tawfiq RK 2025)2.
Trastuzumab Deruxtecan is the world’s first ADC drug targeting HER2 in lung cancer5. This drug employs a highly active drug delivery system with cleavable linker technology, combining a humanized HER2 monoclonal antibody with a toxic small molecule (DXd), possessing the dual advantages of precise targeting and high efficiency with low toxicity. By using cysteine coupling, the drug-antibody ratio (DAR) reaches a theoretical maximum of 8:1. Compared to early developed ADCs, Trastuzumab Deruxtecan exhibits high stability in the bloodstream until internalized by overexpressed peptides in cancer cells, selectively cleaving to release a potent payload that induces DNA damage and apoptosis, and can exert a “bystander” killing effect on adjacent tumor cells.6.
In addition to Trastuzumab Deruxtecan, more ADC drugs targeting lung cancer are making breakthroughs, potentially providing patients with more effective treatment options in the near future.
Challenges Remain, but the Future is Promising—
The Development Direction of ADC Drugs in Lung Cancer
The future of ADC drugs is evolving along a clear trajectory, but existing challenges need to be addressed. For instance, tumor antigen expression heterogeneity may lead to inconsistent responses and treatment outcomes. Other significant challenges include adverse reactions and treatment resistance.2 Therefore, the development direction of ADC drugs in lung cancer mainly focuses on the following aspects:
Developing New Targets
In the selection of targets for ADC drugs, in addition to tumor cell surface targets, targeting the tumor microenvironment is a new hotspot in this field. Such targets are more accessible from the bloodstream to ADC molecules as they are distributed around tumor tissues. Besides stronger antitumor effects, the likelihood of developing resistance may also be reduced.1.
From Monotherapy Breakthroughs to Combination Synergy
To improve efficacy and delay resistance, consideration can be given to combining ADCs with other drugs. For example, combining ADCs with immunotherapy or targeted therapy may achieve synergistic or additive effects, thereby enhancing treatment outcomes. For instance, ADC-induced immunogenic cell death can stimulate antitumor immune responses, and immune checkpoint inhibitors may further amplify this immune response.2.
Enhancing Cytotoxic Drug Technology
The efficacy of ADCs is closely related to the number of cytotoxic molecules linked to them; the more cytotoxic molecules linked, the better the efficacy, but this also increases toxicity risks. Conversely, fewer linked cytotoxic molecules may result in insufficient efficacy. Therefore, precisely selecting the number of cytotoxic molecules is a technical challenge. Additionally, choosing the right cytotoxic drug is crucial, balancing its killing ability with its toxicity and efficacy.1.
Conclusion
As a prominent representative of precision medicine, ADC drugs are reshaping the treatment landscape for lung cancer. From the intricate design of the “three-body” structure to remarkable clinical achievements, and to the diverse explorations of future trends, ADC drugs not only break through the efficacy bottlenecks of traditional treatments but also open a new era of precision medicine for lung cancer. With technological iterations and strategic innovations, ADC drugs are expected to transition from monotherapy to combination therapy, from late-line to frontline, bringing hope for longer survival and higher quality of life for lung cancer patients, illuminating the path for humanity to conquer cancer.
References:
1. Liu Wenchao et al. Current Status and Prospects of Antibody-Drug Conjugates. Progress in Biochemistry and Biophysics. 2023;50(5):1167-1189.
2. Tawfiq RK et al. Targeting Lung Cancer with Precision: The ADC Therapeutic Revolution. Current Oncology Reports. https://doi.org/10.1007/s11912-025-01655-5
3. Fu ZW et al. Antibody Drug Conjugate: The “Biological Missile” for Targeted Cancer Therapy. Signal Transduction and Targeted Therapy 2022; 7:93.
4. Staudacher AH et al. Antibody Drug Conjugates and Bystander Killing: Is Antigen-Dependent Internalization Required? British Journal of Cancer 2017; 117:1736–1742.
5. Mehta GU et al. FDA Approval Summary: Fam-Trastuzumab Deruxtecan-nxki for Unresectable or Metastatic Non-Small Cell Lung Cancer with Activating HER2 Mutations. The Oncologist, 2024, 29, 667–671.
6. Li BT et al. Trastuzumab Deruxtecan in HER2-Mutant Non-Small-Cell Lung Cancer. N Engl J Med. 2022 Jan 20;386(3):241-251.
Approval Code: CN-20250606-00008
