
First Author: Zhou Tong, Luo Zhongge, Zhao Jiangting
Corresponding Authors: Liu Qingju (Yunnan University, e-mail:[email protected])
Tang Junwang (Tsinghua University, e-mail: [email protected])
Abstract:Photocatalytic hydrogen production is considered a promising strategy to mitigate the environmental impact of carbon-intensive chemical industries. Previous studies have shown that loading single atoms can significantly enhance photocatalytic efficiency, and their catalytic activity is largely influenced by the microenvironment. Therefore, achieving precise control over the microenvironment of single atoms is crucial, yet it remains a significant challenge. This study reports a novel Pt-C/TiO2 photocatalyst, which simultaneously loads Pt quantum dots (PtQD) and carbon-coordinated Pt single atoms (PtSA). Under experimental conditions, the hydrogen production rate of this photocatalyst reached 43.2 mmol·h-1. Notably, the number of hydrogen molecules generated per unit incident photon (H2/photon) reached as high as 0.92. The unique Pt-C coordination environment not only provides a direct transport pathway for photogenerated electrons but also activates the surrounding Ti, effectively promoting the separation of electron-hole pairs and enhancing hydrogen production performance.
Introduction:Methanol solution not only has the advantages of low cost and convenient transportation but is also an ideal hydrogen storage medium (with a hydrogen content of up to 12.6 wt%), thus attracting widespread attention in recent years, especially in applications combined with proton exchange membrane fuel cells (PEMFC). However, the traditional methanol solution hydrogen production process usually requires temperatures above 200 °C and pressure conditions of 25–50 bar. Additionally, the generation of carbon monoxide (CO) during the reaction process is inevitable, which significantly limits its application and promotion.
In recent years, the photocatalytic hydrogen production pathway coupled with the generation of formaldehyde (HCHO) in methanol solution has received increasing attention. This method not only provides an alternative pathway for traditional industrial formaldehyde production but also offers a new solution for the low-cost synthesis and separation of hydrogen. Previous studies have shown that the synergy between metal single atoms and metal quantum dots can not only maintain the high dispersion of active sites on the photocatalyst surface but also exhibit excellent selectivity for formaldehyde in methanol dehydrogenation reactions. Further research indicates that the local coordination environment formed between single atom metals and supports significantly affects the electronic structure of the active centers, thereby determining their catalytic efficiency. Therefore, regulating the coordination environment of single atom metals is considered an effective strategy to optimize their electronic structure and enhance catalytic performance.
Titanium dioxide (TiO2) is a widely studied photocatalyst, and its performance can be further enhanced by loading single atom catalysts (SACs), such as platinum single atoms (PtSA). In the currently reported TiO2 supported PtSA system, Pt atoms mainly exist in the first coordination shell in the form of Pt–O, which helps to achieve uniform dispersion on the substrate surface, but the efficiency of photocatalytic hydrogen production still fails to meet practical application requirements.
 Figure1. Characterization of the morphology and structure of the photocatalyst.(A) Scanning electron microscope (SEM) image of TiO2. (B) High-resolution transmission electron microscope (HRTEM) lattice image of Pt–C/TiO2. (C) High-angle annular dark field scanning transmission electron microscope (HAADF-STEM) image (Pt quantum dot particle size distribution of 1–5 nm). (D) Elemental mapping of Pt–C/TiO2. (E–F) HAADF-STEM images.(G) Corresponding line scan analysis spectrum marked in (F), Line 1 and Line 2 show the distribution of Pt, Ti and Ti vacancies.
Figure1. Characterization of the morphology and structure of the photocatalyst.(A) Scanning electron microscope (SEM) image of TiO2. (B) High-resolution transmission electron microscope (HRTEM) lattice image of Pt–C/TiO2. (C) High-angle annular dark field scanning transmission electron microscope (HAADF-STEM) image (Pt quantum dot particle size distribution of 1–5 nm). (D) Elemental mapping of Pt–C/TiO2. (E–F) HAADF-STEM images.(G) Corresponding line scan analysis spectrum marked in (F), Line 1 and Line 2 show the distribution of Pt, Ti and Ti vacancies.
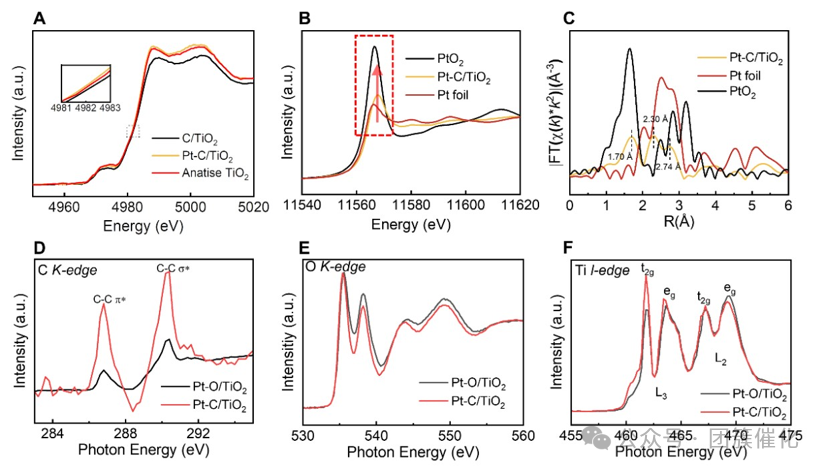
Figure2. Chemical characterization of the catalyst.(A) TiO2, C/TiO2 and Pt–C/TiO2 Ti K edge XANES spectra.(B) Pt–C/TiO2, PtO2 and Pt foil Pt L edge XANES spectra.(C) Pt–C/TiO2, PtO2 and Pt foil Fourier transform results of the k2 weighted EXAFS spectra at the Pt L edge.(D) Pt–O/TiO2 (black curve) and Pt–C/TiO2 (red curve) normalized C K edge spectra.(E) Pt–O/TiO2 (black curve) and Pt–C/TiO2 (red curve) normalized O K edge spectra.(F) Pt–O/TiO2 (black line) and Pt–C/TiO2 (red curve) normalized Ti L edge spectra.
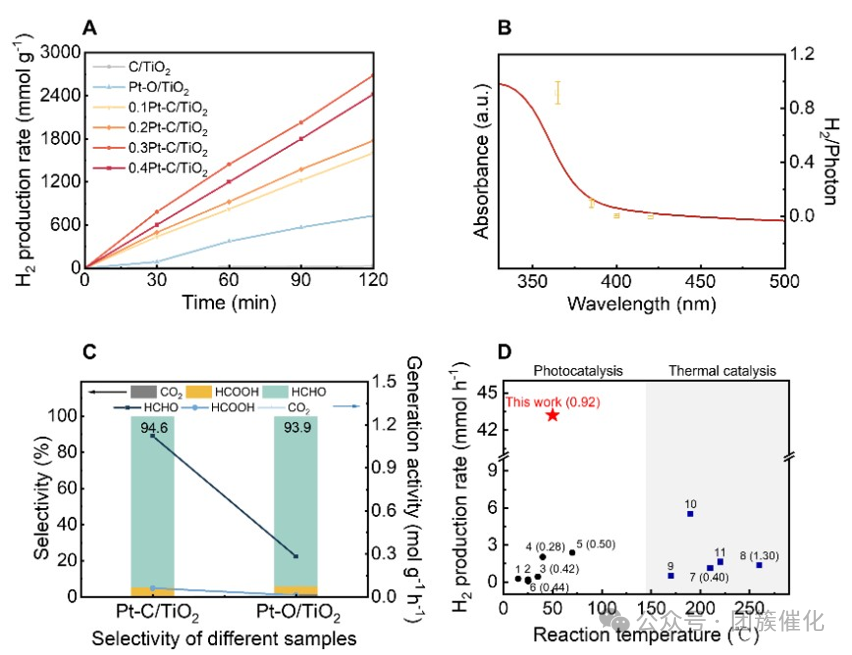
Figure3. Hydrogen production performance of the catalyst.(A) Comparison of catalytic activity of different catalysts at 40 °C, using a 365 nm LED as the light source (light intensity of 344.7 mW/cm2) conditions.(B) The relationship between the number of H2/photon of Pt–C/TiO2 at different wavelengths and its UV-visible absorption spectrum.(C) Comparison of product selectivity of Pt–O/TiO2 and Pt–C/TiO2 photocatalysts.(D) Benchmark comparison of hydrogen production rates of representative photocatalysts and thermal catalysts in methanol production from water solution, where the white background represents photocatalysis and the gray background represents thermal catalysis.

Figure4. Mechanistic analysis and in situ characterization of the catalyst.(A) In situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) of Pt–C/TiO2 under dark and light conditions (300 W xenon lamp, 40 °C).(B) Quasi-in situ XPS Ti 2p photoelectron spectroscopy of Pt–C/TiO2 before and during light exposure.(C) Quasi-in situ XPS Pt 4f photoelectron spectroscopy of Pt–C/TiO2 before and during light exposure.(D) Differential charge density distribution map of Pt–C/TiO2. (E) Energy change curves of two dehydrogenation reaction pathways on Pt–C/TiO2 and Pt–O/TiO2 catalysts during methanol oxidation.
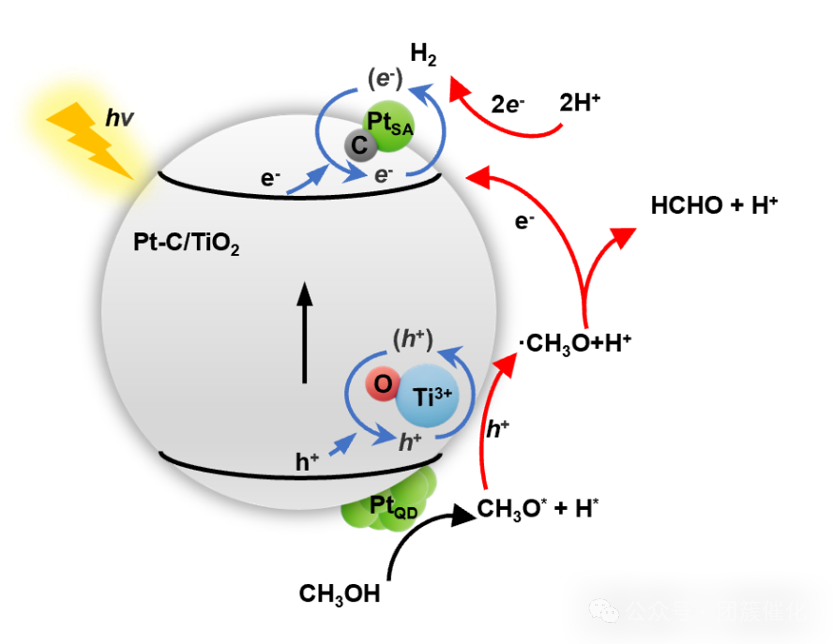
Figure5. Possible reaction mechanism of the catalyst.
Conclusion: This study successfully synthesized a highly efficient Pt–C/TiO2 photocatalyst with a unique Pt–C coordination structure, achieving efficient aqueous methanol hydrogen production under ambient conditions. In this catalyst, the doped carbon atoms occupy the Ti vacancies and surrounding oxygen sites, forming a coordination structure with Pt single atoms (PtSA). Under the conditions of 365 nm LED light and 40 °C, the optimized Pt–C/TiO2 photocatalyst can achieve a hydrogen generation rate of 43.2 mmol·h-1 at a dosage of 70 mg; with a catalyst usage of 10 mg, its normalized hydrogen production rate reaches as high as 1.34 mol·g-1·h-1. Meanwhile, the Pt content in this catalyst is only 0.820 wt.%; its hydrogen production process exhibits excellent stability, and no carbon monoxide was detected in the products. Further research results show that the pre-activation effect of Pt quantum dots (PtQD) on methanol molecules, the efficient photogenerated electron transport pathway formed by the coordination of PtSA with carbon, and the abundant active sites provided by PtSA and Ti3+ collectively promote the enhancement of the photocatalytic hydrogen production rate.
Article Information:Synergy between unique Pt-C coordination and Pt quantum dots on TiO2 for exceptional photocatalytic methanol dehydrogenation. Science Advances, 2025, 11, eadw2028. https://doi.org/10.1126/sciadv.adw2028.