

【Research Background】
Replacing graphite anodes with lithium metal anodes (theoretical specific capacity of 3860 mA h g–1) is considered one of the most attractive ways to improve battery energy density.
The electrolyte plays an important role in insulating electrons but conducting ions between the electrodes, thus promoting battery performance. With the rapid development of electrochemical energy storage technology, traditional electrolytes can no longer meet the requirements of high energy density batteries. Furthermore, lithium metal, as the ultimate anode material, poses high demands on the electrolyte. Safety hazards arising from lithium dendrite growth and flammable electrolytes are major issues limiting the practical application of lithium metal anodes. In addition to optimizing traditional electrolytes, various electrolyte design concepts have been proposed, including ultra-concentrated, polymer, solid-state, ionic liquid, and eutectic electrolytes. Among these conceptual electrolytes, eutectic electrolytes are a branch of deep eutectic solvents, typically prepared from a eutectic mixture of Lewis or Brønsted acids and bases. The intermolecular interactions between different components lead to a low melting point. Eutectic electrolytes generally consist of metal salts and hydrogen bond donors, facilitating advancements in the field of metal battery applications. Eutectic electrolytes have attracted significant research interest due to their non-toxic, low-cost, and environmentally friendly properties compared to organic carbonates and ionic liquid electrolytes (Figure 1a). However, research on eutectic electrolytes is still in its early stages. Despite many advantages, high viscosity and low ionic conductivity are major issues limiting their application in energy storage.

Figure 1. (a) Radar chart of different electrolyte characteristics, where the highest values represent the best characteristics. (b) Solvation shell dominated by BL, (c) Solvation shell dominated by SN, and (d) Schematic of competitive solvation shell.
【Work Summary】
Recently, Professors Li Jie Ci, Mingyu Li, and Jiaheng Zhang from Harbin Institute of Technology (Shenzhen) and their team designed a ternary eutectic electrolyte composed of LiTFSI (TFSI = bis(trifluoromethanesulfonyl)imide), butyrolactam (BL), and succinonitrile (SN). This electrolyte exhibits high ionic conductivity, non-flammability, and a wide electrochemical window.
Additionally, a “competitive solvation” mechanism between SN and BL has been proposed to understand the improved characteristics of the ternary eutectic electrolyte (Figure 1b-d). The competitive solvation effect between SN, BL, and Li+ reduces viscosity and enhances the stability of the eutectic electrolyte. The preferential coordination of BL to Li+ facilitates the formation of a stable solid electrolyte interphase (SEI) membrane, leading to uniform and dendrite-free Li plating. Benefiting from the advantageous properties of the ternary eutectic electrolyte, LiFePO4 (LFP)||Li batteries demonstrate excellent cycling performance and higher Coulombic efficiency, even under harsh conditions such as high current density, low temperature, and high temperature.
Using this ternary eutectic electrolyte, LiFePO4/Li batteries maintain a capacity retention rate of up to 90% after 500 cycles at 2C, with an average Coulombic efficiency of 99.8%. Nickel-rich LiNi0.8 Co0.1 Al0.1 O2/Li and LiNi0.8 Co0.1 Mn0.1 O2/Li batteries based on modified ternary eutectic electrolytes achieve excellent cycling performance. This study provides insights for understanding and designing better electrolytes for lithium metal batteries and similar sodium/potassium metal batteries.
This achievement was published in the top international journal “A Competitive Solvation of Ternary Eutectic Electrolytes Tailoring the Electrode/Electrolyte Interphase for Lithium Metal Batteries” in ACS Nano (IF 18.027) with Wanbao Wu as the first author.
【Detailed Content】
Physical and Chemical Properties of Ternary Eutectic Electrolytes
The physical and chemical properties of the eutectic electrolyte are displayed in Figure 2b. In the binary eutectic electrolyte, LB (molar ratio of LITFSI to BL is 1:3) shows an ionic conductivity of 0.43 mS cm–1 and a high viscosity of 238.1 mPa s. In comparison, the LS electrolyte composed of LiTFSI and SN with a molar ratio of 1:4 exhibits a high ionic conductivity of 1.72 mS cm–1 and a relatively low viscosity of 86.0 mPa s. For the ternary eutectic electrolyte, the viscosity decreases with increasing SN content. When the molar ratio of SN increases from 1 to 5, the viscosity significantly decreases from 137.6 mPa s to 27.5 mPa s, while the conductivity increases from 1.25 to 2.83 mS cm–1. Notably, at a molar ratio of 5 for SN, the lithium salt concentration in the electrolyte is only 1.15M, which is close to the commercial electrolyte concentration of 1M. Moreover, LB and LiTFSI, BL, and SN (LBS) with fluoroethylene carbonate (FEC) as an additive can form a stable SEI membrane to prevent further reaction of BL with metallic lithium.
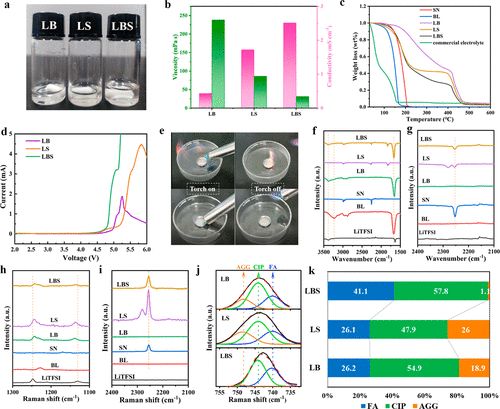
Figure 2. Comparison of properties of different electrolytes and the physical and chemical properties of eutectic electrolytes.
The thermal stability of the electrolyte is of considerable importance for battery safety in cases of short circuits or thermal runaway. As shown in Figure 2c, pure BL loses 50.45% of its weight at 150°C. However, only a 3.06% weight loss is observed in LB, due to the strong interaction between LiTFSI and BL after forming the eutectic electrolyte, which suppresses the decomposition of BL. Pure SN only loses 12.09% at 150°C; thus, LS exhibits good thermal stability, losing 13.73%. For the ternary eutectic electrolyte, all electrolytes lose about 10% of their weight at 150°C. In contrast, the commercial carbonate electrolyte loses 86.26% of its weight at 150°C. The good thermal stability of ternary eutectic electrolytes can be attributed to the interactions between LiTFSI, BL, and SN, as well as the inherent thermal stability of SN. When the molar ratio of SN is 4, the lowest eutectic temperature of the ternary eutectic electrolyte is observed at -86.95°C, which increases to -73.14°C when the molar ratio of SN is reduced to 1. The eutectic temperature of the ternary eutectic electrolyte is much lower than that of the binary eutectic electrolyte LB by 1.7°C. Notably, when the molar ratio of SN increases to 5, two crystallization peaks appear at -46.53°C and -84.43°C, which may be due to the phase change of SN. The electrochemical stability of binary and ternary eutectic electrolytes was tested using LSV.
As shown in Figure 2d, the results indicate that the oxidation resistance of these electrolytes is higher than 4.5V. This suggests compatibility with most lithium battery systems, including high-voltage cathode materials. Simultaneously, aluminum current collectors can be passivated in ternary eutectic electrolytes. Considering the physical and chemical properties and cost factors, the ternary eutectic electrolyte with a molar ratio of 4 for SN is selected as a representative, defined as LBS. For comparison, binary eutectic electrolytes LB and LS, as well as a commercial electrolyte, were also studied.
Flammability tests were conducted on commercial electrolytes and ternary eutectic electrolytes (Figure 2e). Commercial electrolytes are easily ignited, posing significant safety risks to batteries in practical applications. In contrast, ternary eutectic electrolytes do not support combustion, indicating good non-flammability and safety.
Solvation Structure Analysis of Ternary Eutectic Electrolytes
The formation mechanism of ternary eutectic electrolytes was studied using FTIR and Raman spectroscopy. Competitive solvation exists between BL and SN with Li+, where BL preferentially forms solvation structures with Li+, as illustrated in Figure 1d. Furthermore, Figure 1b shows the solvation structure of LB without SN, indicating that BL participates in the first solvation shell. The various bonds of the original components are collectively weakened by the intermolecular interactions among LiTFSI, BL, and SN, leading to a eutectic solution. (Figure 2a). The competitive solvation in ternary eutectic electrolytes results in low viscosity, high ionic conductivity, and stable electrolytes.
Analysis of Lithium Deposition Morphology and Theoretical Simulation of Competitive Solvation
The compatibility of different electrolytes with lithium metal anodes was assessed through cycling Li||Li symmetric cells (Figure 3a). The ternary eutectic electrolyte with competitive solvation can effectively regulate morphology, achieving dendrite-free lithium deposition. Compared to binary eutectic electrolytes, using ternary eutectic electrolytes yields more stable cycling performance for lithium-ion symmetric cells.
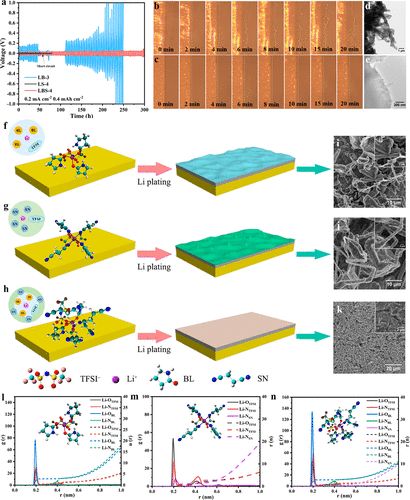
Figure 3. Electrochemical symmetric battery tests of different eutectic electrolytes, lithium deposition morphology, and theoretical calculations.
Theoretical simulations can better understand the competitive solvation mechanism and ionic specifications of ternary eutectic electrolytes. According to Figure 3l-n, the radial distribution functions and coordination numbers of LB, LS, and LBS show that the first Li+ coordination shell (within 2.80 Å) in LB is primarily dominated by Li-OBL and Li-OTFSI at 1.93 Å, with coordination numbers of 2.85 and 1.08, respectively. For LS, peaks in the radial distribution function (RDF) at 1.93 Å and 2.16 Å are clearly observed, attributed to Li-OTFSI and Li-NSN. This indicates that SN molecules participate in the solvation structure of Li+. By adding SN to form the ternary eutectic electrolyte (LBS), the RDF peak corresponding to the Li-OBL coordination structure significantly increases at 1.93 Å as SN content increases. This suggests enhanced coordination between Li+ and BL. (31) For LB and LS, each Li+-solvation shell often coordinates with two or three TFSI– anions. In contrast, for LBS, only one TFSI– anion (on average) is observed in the Li+-solvation shell. (Additionally, the coordination number of Li-OTFSI decreases from 1.08 in LB to 0.93 in LBS, indicating that the introduction of SN promotes the dissociation of Li+ and TFSI–. Notably, no RDF peak for Li-NSN is observed in the ternary eutectic electrolyte. Furthermore, the coordination number of Li-NSN significantly decreases from 2.11 in LS to 0.04 in LBS, indicating that SN does not participate in the solvation shell. These MD results corroborate with FTIR and Raman characterization results, further confirming the existence of competitive solvation structures in ternary eutectic electrolytes.
In the case of LB, Li+ coordinates with the C═O of BL and the S═O of TFSI– anions, and as the amount of BL increases, Li+ preferentially coordinates with BL, facilitating the dissociation of Li+ and TFSI– anions. For the LS binary system, the formation of Li-N (approximately 2 Å) between Li+ and SN can be observed, even as the number of SN increases to 4. This is consistent with the observation of solvation structures involving coordination between LiTFSI and SN in LS using Raman spectroscopy. Furthermore, the coordination of LiTFSI with different amounts of BL and SN in ternary eutectic electrolytes is verified, where Li+ preferentially coordinates with BL in the presence of SN, especially when the amount of BL exceeds 3. This validates that SN does not participate in the solvation structure. Reduced Density Gradient (RDG) analysis shows weak intermolecular interactions between LiTFSI, BL, and SN. ESP analysis of the electronic density of LBS shows that the negative electronegative regions of the O atoms in BL and the N atoms in SN suggest a tendency to coordinate with the positive charge regions around Li+, leading to a stable eutectic structure. LiTFSI(BL)3 (SN)2 structure presents the lowest potential energy and a more uniform distribution of molecular electrostatic potential energy surface, indicating a high tendency of this coordination structure. This competitive solvation structure plays a crucial role in the formation of the solid electrolyte interphase (SEI) membrane and electrolyte stability. Species coordinated with Li+ ions are strongly polarized and preferentially reduced, forming the main components of the SEI membrane. Therefore, in ternary eutectic electrolytes, the strong interaction of BL and TFSI– with Li+ largely determines the composition and structure of the SEI membrane. In contrast, SN does not participate in the solvation structure, reducing spontaneous chemical reactions between SN and metallic Li. Thus, the addition of SN reduces the viscosity of the electrolyte and enhances ionic conductivity.
Electrochemical Performance of Batteries

Figure 4. Electrochemical performance of LFP/Li batteries with different electrolytes: (a) Rate performance of LFP/Li batteries using LBS and commercial electrolytes at 25°C. Cycling performance and corresponding Coulombic efficiency of LFP/Li batteries using different electrolytes at 25°C (b) at 1 C, (c) at 2 C, and (d) at 5 C. (e) Low-temperature performance of LFP/Li batteries using LBS at 0 C and 0.1 C. (g) Cycling performance and corresponding Coulombic efficiency of high-load LFP/thin Li full batteries using LBS electrolyte at 0.5C, 25°C.

Figure 5. Electrochemical performance of NCA/Li batteries with different electrolytes. (a) Rate performance of NCA/Li batteries with different electrolytes at 25°C. (b) Cycling performance and corresponding Coulombic efficiency of NCA/Li batteries at 1°C. (c) Cycling performance of NCM811/Li batteries at 0.2°C. (d) Electrochemical impedance spectroscopy results of NCA/Li batteries using (d) LBS and (e) LBS-M during different cycles.
Analysis of the Interface Between Electrode and Electrolyte
X-ray photoelectron spectroscopy (XPS) analysis was performed on the NCA cathode during cycling to understand why LBS-M electrolyte exhibits better cycling stability than LBS electrolyte. As shown in Figure 6a, the presence of LiF in the F 1s spectrum indicates the decomposition of LiTFSI at high charging voltages. Additionally, the presence of B-F and B-O in LBS-M indicates that LiDFOB participates in the formation of the CEI layer (Figure 6c). Specifically, DFOB– anions gain electrons during CEI formation and are reduced to B-O bonds and oxalic rings, generating oxygen and -BF2 free radicals, forming a CEI membrane rich in B-O and B-F. The presence of F– and B– species can facilitate the formation of a dense and thin CEI layer, which can suppress the dissolution of transition metal ions and reduce side reactions of the electrolyte. For the O 1s spectrum in Figure 6b, no signals of metal oxide bonds (M-O) were observed at approximately 530 eV for both electrolytes, indicating that ternary eutectic electrolytes can effectively passivate the highly active cathode surface. The effective protection of the NCA cathode by the LiDFOB additive is confirmed by TEM. After cycling 10 times in both ternary eutectic electrolytes, CEI layers of different thicknesses were observed. The thickness of LBS electrolyte is approximately 86 nm (Figure 6d), while that of LBS-M electrolyte is approximately 32 nm (Figure 6e). The results show that the modified ternary eutectic electrolyte effectively protects the NCA cathode, thereby reducing further decomposition of the electrolyte during cycling.

Figure 6. Characterization of CEI components on NCA cathodes cycled in LBS (top) and LBS-M (bottom).
The cycling stability of the battery is determined not only by the electrolyte stability on the cathode surface but also by the electrolyte on the lithium metal anode. Morphological characterization of metallic lithium anodes after 100 cycles in the two ternary eutectic electrolytes was performed using SEM (Figure 6f,g). The anode surface in LBS electrolyte is loose and porous, directly exposing bulk lithium to the liquid electrolyte, leading to continuous corrosion of the lithium metal anode. The morphology of the NCA cathode surface and separator after 100 cycles also confirms severe side reactions in NCA/Li batteries in LBS electrolyte. These results explain the rapid capacity decay of the battery in LBS, as shown in Figure 5b. In contrast, a smooth, flat, and uniform lithium metal anode surface is clearly observed in LBS-M (Figure 6g). This indicates that the surface layer can effectively protect the metallic lithium anode from continuous corrosion by the electrolyte. These results are consistent with the atomic force microscopy (AFM) images in Figure 6h,i, confirming that the metal lithium surface cycled in LBS-M is smoother than that cycled in LBS.
A stable SEI membrane is crucial for excellent cycling performance, which is studied with XPS spectra of the cycled lithium anode (Figure 6j-n). In the F 1s spectrum, the presence of LiF (685 eV) can be observed in both electrolytes (Figure 6l), which comes from the reduction of FEC or fluorinated TFSI– and DFOB– anions. Although LiF has low ionic conductivity, its low solubility in the solvent and high Young’s modulus contribute significantly to the stability of the SEI film. In LBS-M, B-O and B-F peaks are clearly detected, as the bond energy of B-O is much weaker than that of other bonds (Figure 6n). Therefore, the B-O bond is easily broken, and the reaction between LiDFOB and metallic lithium participates in the formation of the SEI membrane. In the C 1s spectrum (Figure 6j), the intensity of the C-F peak generated by the decomposition of LiTFSI in LBS-M is stronger than that in LBS, indicating more decomposition of LiTFSI to form the SEI membrane in the presence of LiDFOB. Notably, in both ternary eutectic electrolytes, Lix N with high ionic conductivity appears, which may facilitate the rapid migration of Li+ and lead to uniform lithium deposition (Figure 6m). The modified ternary eutectic electrolyte not only plays a key role in regulating SEI and CEI components but also improves compatibility with lithium metal anodes (Figure 7). The stable SEI membrane and the good compatibility with metallic lithium contribute to its excellent performance.

Figure 7. Schematic of competitive solvation and regulation of SEI and CEI in ternary eutectic electrolytes.
【Conclusion】
A non-flammable ternary eutectic electrolyte has been developed, which improves the cycling performance and safety of lithium metal batteries through competitive solvation structures with lithium ions. The ternary eutectic electrolyte significantly improves LMB performance in three aspects.
(1) The competitive solvation mechanism reduces side reactions between SN and metallic lithium and the viscosity of the electrolyte while enhancing the stability of the electrolyte.
(2) A stable SEI membrane with LiF and nitrogen-rich species leads to uniform and dense lithium deposition.
(3) The modified ternary eutectic electrolyte effectively enhances the cathode-electrolyte interface.
Thanks to these advantages of the ternary eutectic electrolyte, both LFP/Li and NCA/Li batteries can significantly extend their cycle life and improve Coulombic efficiency. This study provides an in-depth investigation into the competitive solvation of electrolytes. This strategy is promising and highly desirable for developing highly safe and durable lithium metal batteries, as it can benefit from the formation of ternary eutectic electrolytes with reduced lithium salt concentration and viscosity.
A Competitive Solvation of Ternary Eutectic Electrolytes Tailoring the Electrode/Electrolyte Interphase for Lithium Metal BatteriesACS Nano ( IF 18.027 ) Pub Date : 2022-08-30 , DOI: 10.1021/acsnano.2c05016Wanbao Wu, Yihong Liang, Deping Li, Yiyang Bo , Dong Wu, Lijie Ci, Mingyu Li, Jiaheng Zhang
