Skip to content
Electrocatalytic conversion of carbon dioxide (CO2) into useful fuels and chemical feedstocks is an emerging approach to alleviate global warming and reduce reliance on fossil fuels. Methanol (CH3OH), as one of the most important and widely used liquid fuels generated through CO2 reduction, is crucial in the chemical industry.
In this review, we reveal the mechanism of selective generation of CH3OH through CO2 reduction, including catalyst composition design, local structure regulation, and electrolyte/catalyst interface modulation.Finally, we discuss the challenges and future prospects of CO2 reduction to produce CH3OH.
01 Research Background and Objectives
Objective: To explore the origin of catalytic selectivity in the electrochemical conversion of CO2 to methanol, aiming to develop efficient catalysts and promote the effective utilization of CO2.
As the issue of global warming becomes increasingly severe, reducing CO2 emissions and seeking renewable energy alternatives to fossil fuels have become research hotspots. Methanol, as an important liquid fuel and chemical feedstock generated from CO2 reduction, has broad application prospects. However, the catalytic selectivity of this process still faces challenges.
Specific Methods: Advanced electrocatalytic technologies were employed to design and prepare catalysts with specific structures, studying the reaction mechanisms at the electrolyte-catalyst interface to reveal the origin of catalytic selectivity.
Experimental Details: The performance of catalysts was evaluated through experimental methods such as electrochemical testing and catalyst characterization, exploring the relationship between their structure and performance.
Main Findings: The influence of catalyst structure on the catalytic selectivity of CO2 reduction to methanol was revealed, clarifying the important roles of reaction pathways and interface effects in the catalytic process.
Specific Data: Comparative analysis of the performance of different catalysts showed that those with specific structures exhibited higher catalytic selectivity and activity in methanol production.

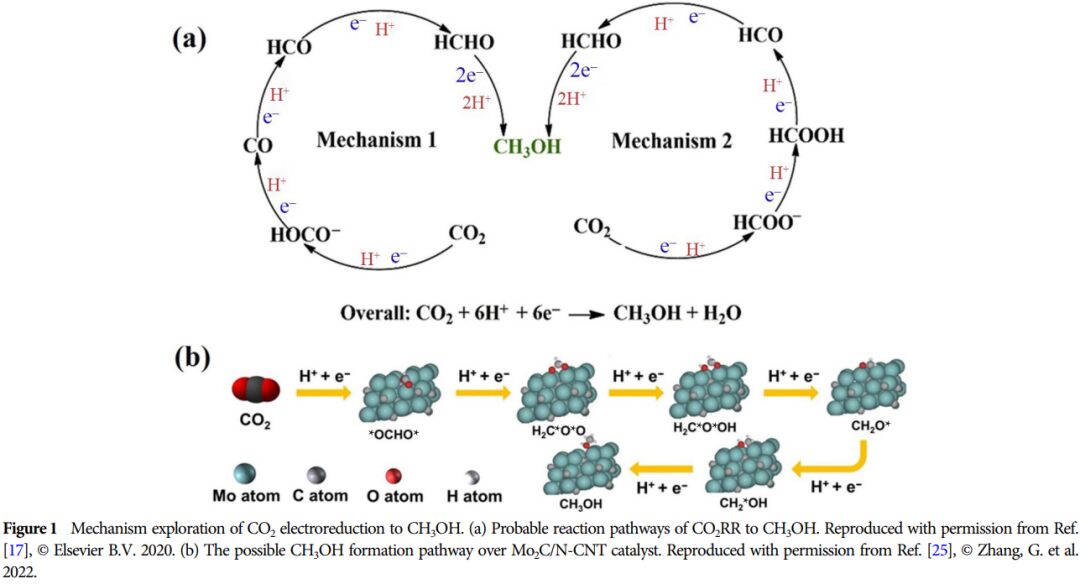
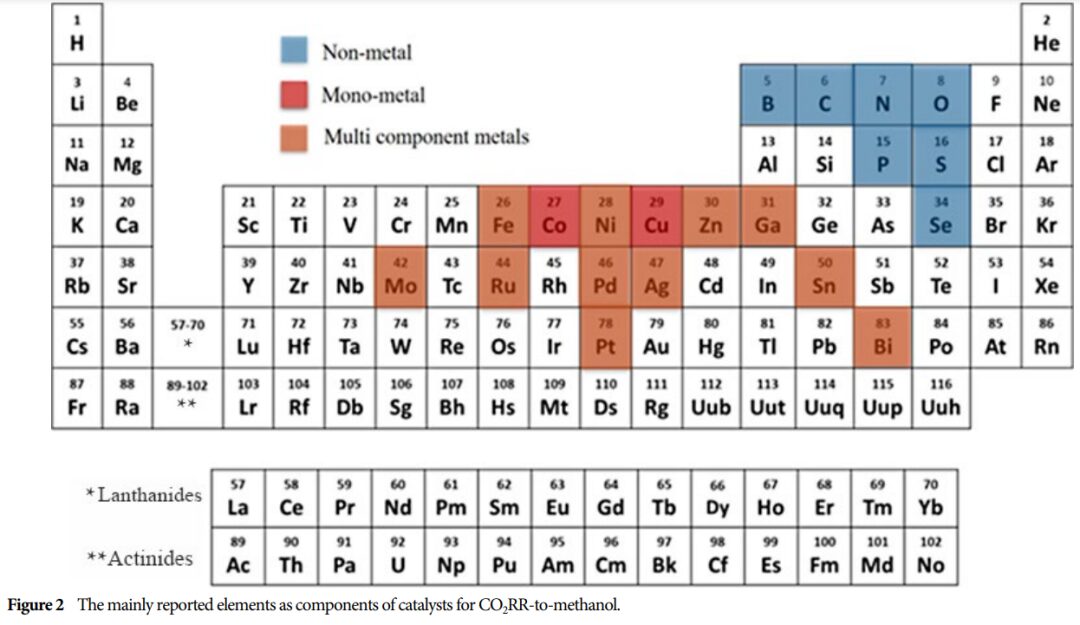
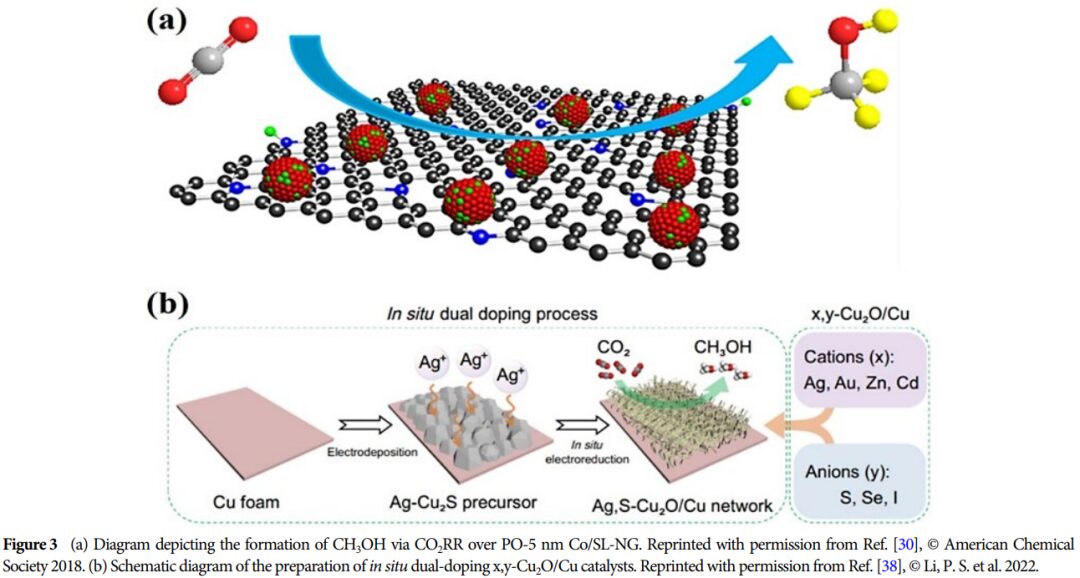
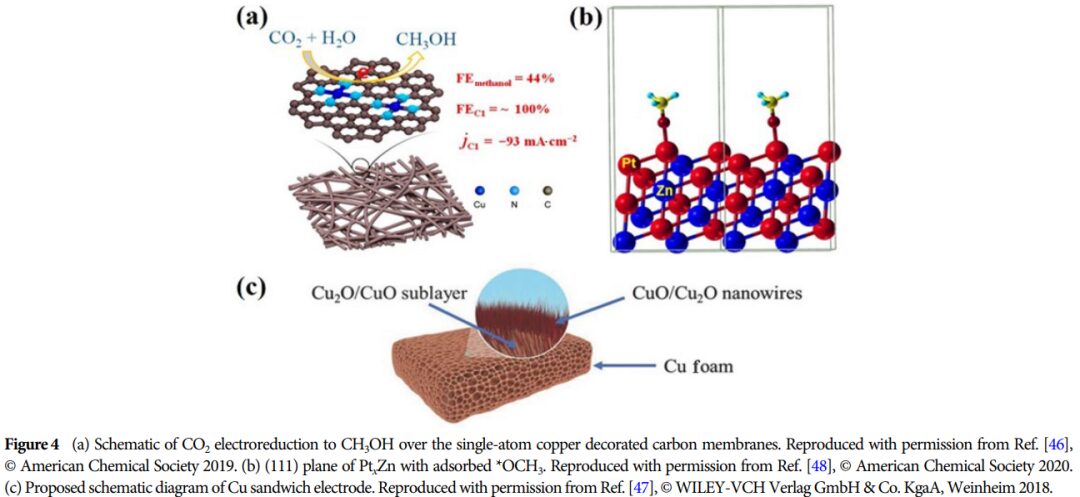

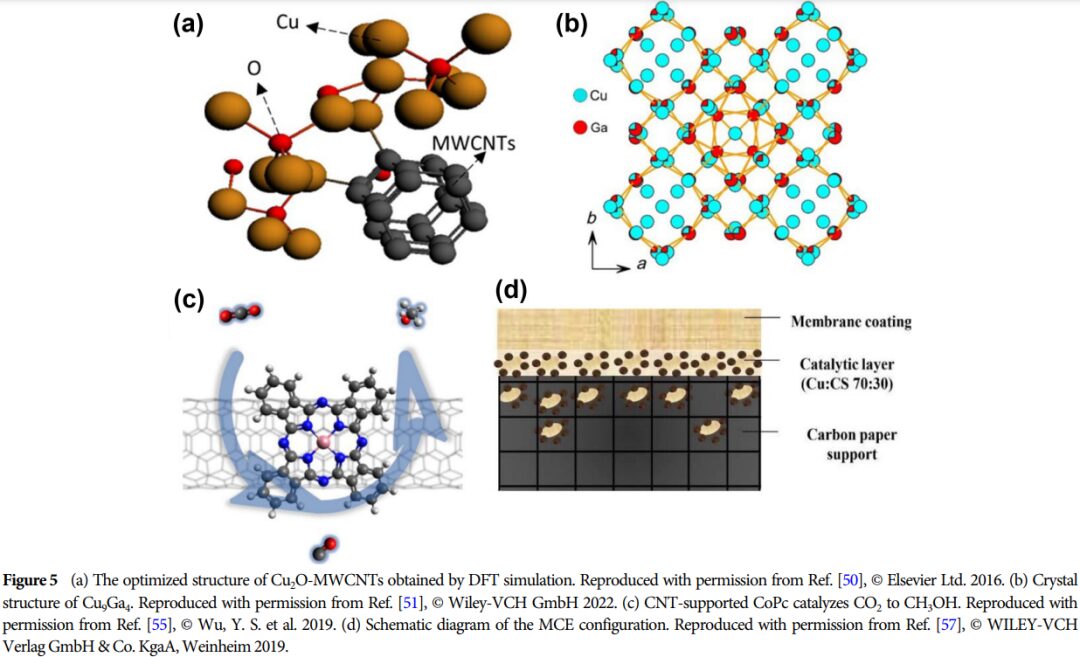
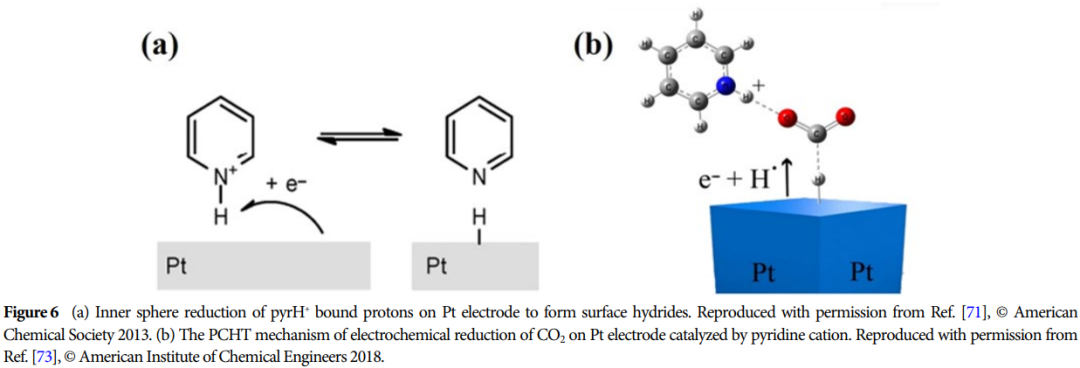
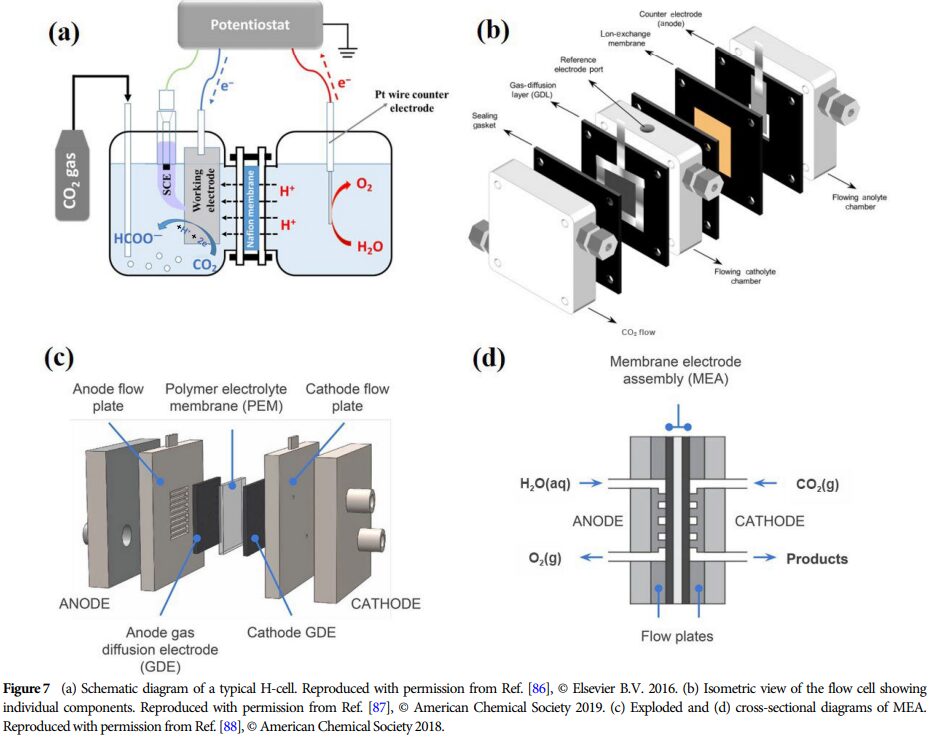
Importance: This research provides a theoretical basis for developing efficient catalysts and promoting the effective utilization of CO2, which is of great significance for advancing energy conversion and environmental protection.
Application Prospects: The research results are expected to be applied in the field of CO2 electrocatalytic conversion, providing new pathways for the production of methanol and other chemical feedstocks.
Limiting Factors: Despite certain progress, the stability of catalysts, optimization of reaction conditions, and cost issues remain key factors limiting the large-scale application of CO2 electrocatalytic conversion technology.
Improvement Directions:Future efforts should further explore more stable catalyst materials, optimize reaction conditions, and reduce costs to enhance the economic viability and feasibility of the technology.
07 Future Research Directions
Research Trends: Future research will focus on developing new catalysts, deeply exploring reaction mechanisms, and promoting system integration to achieve breakthroughs and advancements in CO2 electrocatalytic conversion technology.
Outlook: As research continues to deepen, CO2 electrocatalytic conversion technology is expected to make greater contributions to alleviating global warming and promoting sustainable development.
In summary, this study delves into the origin of catalytic selectivity in the electrochemical conversion of CO2 to methanol, achieving significant progress while still facing some challenges and limitations. Future research needs to further explore new catalysts, optimize reaction conditions, and promote system integration to realize breakthroughs and widespread applications of this technology.
▲ Disc Electrode Spin Coater





















