Click the blue text
Follow us

Author | Bi Rong
Introduction
There are no specialized cells dedicated to specific memories.
——Karl Lashley
We may easily remember a birthday party, the friends who attended, the taste of the cake, and the gifts, but it is difficult to recall what we did the day after the birthday. Everything that people have perceived, thought about, experienced, and acted upon can be retained in the mind in the form of images, which can be reproduced when necessary. This process of retaining and retrieving past experiences in the human brain is called memory [1]. From the perspective of information processing, memory includes three stages: encoding, storage, and retrieval. To make external input information suitable for storage, the brain must process the information finely, first transforming the information from different sensory channels into a form that the human brain can accept; this processing method is called encoding. Encoding of information is equivalent to the recognition process, storage of information is equivalent to the retention process, and retrieval of information is equivalent to the recognition or recall process. Memory is the foundation of all psychological activities; without memory, there would be no perception, experience, concept formation, reasoning, learning, or adaptation to a changing environment.
Based on the duration of retention, memory can be divided into sensory memory, short-term memory, and long-term memory [2]. Sensory memory is similar to perceptual processing and can only last for a few milliseconds to a few seconds. For example, we can remember what someone just said, even if we were not consciously listening at the time. Most information disappears during the sensory memory stage, and only a small portion of information receives attention and enters short-term memory or working memory. Short-term memory can last from a few seconds to a few minutes, such as when we urgently dial a phone number provided by an operator. Subsequently, some important short-term memories are further processed and transformed into long-term memories, which can last from a few days to several years, such as memories of events from childhood. Long-term memory can be divided into declarative memory and non-declarative memory based on the type of stored information. Declarative memory refers to memories that can be accessed through conscious processes, including episodic memory (events that occurred, such as riding a bicycle for the first time) and semantic memory (general knowledge, such as how to do multiplication). Non-declarative memory often lies beyond our conscious reach and includes procedural memory (automated and cognitive skills, such as riding a bicycle/reading), perceptual representation systems, classical conditioning, and non-associative learning. Studies on patients with brain injuries have found that after bilateral temporal lobe resection, sensory memory, short-term memory, and working memory are not damaged, but short-term memory cannot be converted into long-term memory [3].
Working memory is an extension of the concept of short-term memory, representing a limited-capacity process that temporarily holds information and mentally processes it. Its content can originate from sensory memory or be retrieved from long-term memory [4]. Baddeley and others proposed a working memory model, which posits that short-term memory is not a single system but a complex system composed of a central executive system, phonological loop, visuospatial sketchpad, and episodic buffer. The central executive system is the core of working memory, responsible for information control, transformation, and monitoring; the phonological loop and visuospatial sketchpad are responsible for the short-term memory and storage of auditory (acoustic) and visual information, respectively; the episodic buffer is used to store the results of different information processing [5][6]. Different types of memory have different neural mechanisms. Research using positron emission tomography (PET) imaging has found that for verbal working memory, brain activation occurs in the left lateral prefrontal cortex; for spatial working memory, activation mainly appears in the right hemisphere (inferior frontal gyrus, posterior parietal cortex, and occipital lobe) [7]. From human behavior, fMRI decoding, TMS, and electrophysiological studies in monkeys, it has been found that the storage of working memory occurs in the prefrontal cortex (PFC), posterior parietal cortex (PPC), and sensory cortex (as shown in Figure 1) [8].
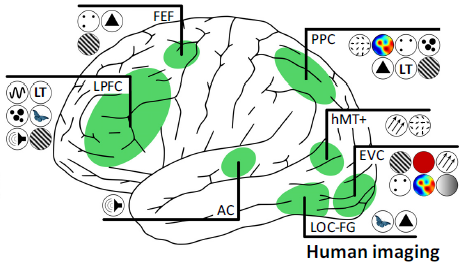
Figure 1 Distribution of working memory content in brain regions
The hippocampus in the medial temporal lobe is a key brain region for processing memory, involved in the encoding, storage, and retrieval of memories. Studies using electrophysiological and molecular biology techniques have found that during memory formation, the amino acids involved in forming long-term potentiation in the hippocampus increase, and learning directly enhances excitatory postsynaptic potentials in the CA1 region of the hippocampus [9]. In the entorhinal cortex connected to the hippocampus, there are neurons that represent spatial locations and are not limited to specific environmental information. When rats move to different spatial locations, these neurons activate, and the activation positions of each neuron combine to form different grid shapes; when location neurons and grid neurons work together, the hippocampus provides precise spatial location and navigation information [10]. Some researchers believe that regardless of the type of memory, the essence of memory is the processing of the relationships between pieces of information [11], and the hippocampal structures can integrate different pieces of information [12]. The left and right hippocampus and the parahippocampal cortex are responsible for processing declarative memory, spatial memory, and environmental information, respectively. Research shows that memory ability is related to the size of the hippocampal structure [13][14], the activation intensity of the hippocampus [15], and the rate of new neuron production in the hippocampus [16][17].
After new memories are encoded in the hippocampus, they are stored through consolidation. Traditional memory theories suggest that consolidation is a one-time process, becoming increasingly stable after consolidation. However, recent studies have found that memory has plasticity, and well-consolidated memories can return to an unstable state, requiring a reconsolidation process. The consolidation and reconsolidation processes work together to promote the stability and updating of memories [18]. During the consolidation process, neurons are reactivated, and studies have found that the neurons involved in processing specific memories are repeatedly activated during and after learning, which can improve memory performance [19]. Research by Nader et al. has demonstrated that activated memories require new proteins to help them reconsolidate; if reconsolidation is disrupted, the memory may be completely destroyed [20]. Simple activation followed by interference is an effective means of disrupting old memories, providing methods to eliminate traumatic, fear-related [21][22], and addictive memories [23].
How do we extract memories stored in the brain? The context reinstatement hypothesis suggests that recall involves the process of orienting and reproducing certain representations of stored memories, which can serve as cues to guide and limit the subsequent memory search, thus achieving memory retrieval. Early studies have shown that the retrieval phase reactivates the sensory cortex used during encoding [24], indicating that memory retrieval may involve the re-presentation of information encoded earlier. Research by Polyn et al. [25] has shown that during the memory retrieval phase, category-specific neural activation patterns are re-presented. Some studies have used the similarity of representations during encoding and retrieval as an indicator of neural activation pattern re-presentation, finding that successful recall during the retrieval phase is accompanied by the re-presentation of item-specific neural activation patterns in the parahippocampal cortex (PHC) [26]. Other studies have found that category-specific neural activation pattern re-presentation is distributed in the ventral visual area and frontal-parietal cortex, while item-specific neural activation pattern re-presentation is distributed in the angular gyrus [27].
So how can we improve learning and memory? A universally applicable method is repeated learning, commonly known as “practice makes perfect.” The effectiveness of repeated learning is influenced by many factors, the most important of which is the interval between two learning sessions. Ebbinghaus (1885/1964) first proposed the spacing effect, which refers to the phenomenon that memory performance is better when there are intervals between repeated learning items compared to massed learning [28][29]. Studies have found that compared to massed learning, the spaced learning group (with a 25-hour interval) performed better on recognition memory tests two days after scanning. Additionally, the behavioral repetition in spaced learning is smaller, and the neural repetition suppression in the left inferior frontal gyrus (LIFG) and left fusiform gyrus (LFFG) is also lower. Moreover, in spaced learning, participants with lower neural repetition suppression effects in the left inferior frontal gyrus also performed better on recognition memory tests [30]. Research by Xue Gui et al. has also found that better memory is associated with smaller neural repetition suppression in the bilateral fusiform gyrus, and spaced learning can reduce this repetition suppression and improve recognition performance [31]. These studies indicate that spaced learning can reduce neural repetition suppression, thereby enhancing the neural activity of repeated presentations during the encoding phase and leading to better subsequent memory. Additionally, retrieval practice (such as testing) can also improve learning and memory performance [32][33][34], and compared to summarization, keyword memorization, and repeated learning, it is currently the most efficient and widely applicable learning method [35]. An fMRI study required participants to engage in repeated learning for half of the Swahili-English word pairs and retrieval practice for the other half. The results showed that compared to repeated learning, retrieval practice resulted in greater activation in the left inferior frontal gyrus related to semantic processing, the ventral striatum related to motivation, and the midbrain; furthermore, greater activation during retrieval practice in the left parietal and left middle temporal gyrus predicted better memory performance a week later [36]. Another study found that stronger functional connectivity between the hippocampus and the left ventrolateral prefrontal cortex (VLPFC), ventromedial prefrontal cortex (VMPFC), and posterior cingulate cortex during retrieval practice predicted better subsequent memory performance [37]. With more cognitive resources invested and the involvement of the working memory system, retrieval practice is a process of acquiring, processing, integrating, and consolidating semantic relationships, fully engaging cognitive and emotional functions, as well as cortical and subcortical functions, while leveraging the advantages of semantic and contextual memory to enhance learning and memory [38].
Forgetting is a normal process of memory. Early research primarily focused on passive forgetting, aiming to improve memory by reducing forgetting. However, as memory research has entered practical and clinical applications, there has been more attention on active forgetting. Active forgetting refers to the intentional control to specifically disrupt certain memories or prevent their retrieval. Currently, the main methods for disrupting memories are extinction, interference, and suppression. Extinction originates from conditioned reflexes, where researchers repeatedly present neutral stimuli without simultaneously presenting emotional stimuli, allowing participants to gradually learn that the neutral stimuli are safe [39], but its effects are short-lived. Interference disrupts old memories by repeatedly learning new memories [40], but its effects are limited. Suppression is the active process of placing unwanted memories into the unconscious to produce forgetting. fMRI studies have found that the degree of activation in the prefrontal cortex during active suppression is related to the forgetting effects during testing [41][42]. Wang et al. had participants learn both A-C and B-C pairs simultaneously, and after suppressing A-C memories, they found that using either cue A or B could not successfully retrieve C, indicating that suppression affected the target memory itself [43]. Compared to extinction and interference methods, suppression has superior advantages.

The next article will bring you three case studies (Case 1 – The Impact of Working Memory Training on the Brain and Cognitive Abilities; Case 2 – Memory Techniques; Case 3 – Alzheimer’s Disease), allowing everyone to gain a deeper understanding of the memory system in the brain through relevant cases. Stay tuned.
References
[1] Meng Zhaolan. General Psychology [M]. Peking University Press. 1994.
[2] Squire L R. Memory systems of the brain: a brief history and current perspective [J]. Neurobiology of learning and memory, 2004, 82(3): 171-177.
[3] Annese J, Schenker-Ahmed N M, Bartsch H, et al. Postmortem examination of patient HM’s brain based on histological sectioning and digital 3D reconstruction [J]. Nature communications, 2014, 5: 3122.
[4] D’Esposito M, Postle B R. The Cognitive Neuroscience of Working Memory [J]. Annual Review of Psychology, 2015, 66(1):115.
[5] Baddeley A D, Hitch G. Working memory [M]//Psychology of learning and motivation. Academic press, 1974, 8: 47-89.
[6] Baddeley A. The episodic buffer: a new component of working memory? [J]. Trends in Cognitive Sciences, 2000, 4(11):417-423.
[7] Smith E E, Jonides J, Koeppe R A. Dissociating verbal and spatial working memory using PET [J]. Cerebral cortex, 1996, 6(1): 11-20.
[8] Christophel T B, Klink P C, Spitzer B, et al. The Distributed Nature of Working Memory [J]. Trends in Cognitive Sciences, 2017, 21(2):111.
[9] Whitlock J R, Heynen A J, Shuler M G, et al. Learning induces long-term potentiation in the hippocampus [J]. science, 2006, 313(5790): 1093-1097.
[10] Hafting T, Fyhn M, Molden S, et al. Microstructure of a spatial map in the entorhinal cortex [J]. Nature, 2005, 436(7052): 801.
[11] Suzuki W A, Naya Y. The Perirhinal Cortex [J]. Annual Review of Neuroscience, 2014, 37(37):39.
[12] Burgess N, Maguire E A, O’Keefe J. The human hippocampus and spatial and episodic memory [J]. Neuron, 2002, 35(4): 625-641.
[13] Van Petten C. Relationship between hippocampal volume and memory ability in healthy individuals across the lifespan: review and meta-analysis [J]. Neuropsychologia, 2004, 42(10): 1394-1413.
[14] Erickson K I, Voss M W, Prakash R S, et al. Exercise training increases size of hippocampus and improves memory [J]. Proceedings of the National Academy of Sciences, 2011, 108(7): 3017-3022.
[15] Brewer J B, Zhao Z, Desmond J E, et al. Making memories: brain activity that predicts how well visual experience will be remembered [J]. Science, 1998, 281(5380): 1185-1187.
[16] Deng W, Aimone J B, Gage F H. New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory? [J]. Nature reviews neuroscience, 2010, 11(5): 339.
[17] Van Praag H, Kempermann G, Gage F H. Neural consequences of environmental enrichment [J]. Nature Reviews Neuroscience, 2000, 1(3): 191.
[18] Wang Yingying, Zhu Zijian, Wu Yanhong. The dynamic changes of memory: encoding, consolidation, and forgetting [J]. Science Bulletin, 2016, 61(1):12.
[19] Rasch B, Büchel C, Gais S, et al. Odor cues during slow-wave sleep prompt declarative memory consolidation [J]. Science, 2007, 315(5817): 1426-1429.
[20] Nader K, Schafe G E, Le Doux J E. Fear memories require protein synthesis in the amygdala for reconsolidation after retrieval [J]. Nature, 2000, 406(6797): 722.
[21] Schiller D, Monfils M H, Raio C M, et al. Preventing the return of fear in humans using reconsolidation update mechanisms [J]. Nature, 2010, 463(7277): 49.
[22] He J, Sun H Q, Li S X, et al. Effect of conditioned stimulus exposure during slow wave sleep on fear memory extinction in humans [J]. Sleep, 2015, 38(3): 423-431.
[23] Xue Y X, Luo Y X, Wu P, et al. A memory retrieval-extinction procedure to prevent drug craving and relapse [J]. Science, 2012, 336(6078): 241-245.
[24] Wheeler M E, Petersen S E, Buckner R L. Memory’s echo: vivid remembering reactivates sensory-specific cortex [J]. Proceedings of the National Academy of Sciences, 2000, 97(20): 11125-11129.
[25] Polyn S M, Natu V S, Cohen J D, et al. Category-specific cortical activity precedes retrieval during memory search [J]. Science, 2005, 310(5756): 1963-1966.
[26] Staresina B P, Henson R N A, Kriegeskorte N, et al. Episodic reinstatement in the medial temporal lobe [J]. Journal of Neuroscience, 2012, 32(50): 18150-18156.
[27] Kuhl B A, Chun M M. Successful Remembering Elicits Event-Specific Activity Patterns in Lateral Parietal Cortex [J]. Journal of Neuroscience, 2014, 34(23):8051-8060.
[28] Cepeda N J, Pashler H, Vul E, et al. Distributed practice in verbal recall tasks: A review and quantitative synthesis [J]. Psychological bulletin, 2006, 132(3): 354.
[29] Donovan J J, Radosevich D J. A meta-analytic review of the distribution of practice effect: Now you see it, now you don’t [J]. Journal of Applied Psychology, 1999, 84(5): 795.
[30] Wagner A D, Maril A, Schacter D L. Interactions between forms of memory: When priming hinders new episodic learning [J]. Journal of cognitive neuroscience, 2000, 12(Supplement 2): 52-60.
[31] Xue G, Mei L, Chen C, et al. Spaced learning enhances subsequent recognition memory by reducing neural repetition suppression [J]. Journal of cognitive neuroscience, 2011, 23(7): 1624-1633.
[32] Karpicke J D, Roediger H L. The critical importance of retrieval for learning [J]. science, 2008, 319(5865): 966-968.
[33] Karpicke J D, Blunt J R. Retrieval practice produces more learning than elaborative studying with concept mapping [J]. Science, 2011, 331(6018): 772-775.
[34] Tulving E. Episodic memory: from mind to brain. [J]. Rev Neurol, 2004, 53(4 Pt 2):S9.
[35] Dunlosky J, Rawson K A, Marsh E J, et al. Improving students’ learning with effective learning techniques: Promising directions from cognitive and educational psychology [J]. Psychological Science in the Public Interest, 2013, 14(1): 4-58.
[36] van den Broek G S E, Takashima A, Segers E, et al. Neural correlates of testing effects in vocabulary learning [J]. Neuroimage, 2013, 78: 94-102.
[37] Wing E A, Marsh E J, Cabeza R. Neural correlates of retrieval-based memory enhancement: an fMRI study of the testing effect [J]. Neuropsychologia, 2013, 51(12): 2360-2370.
[38] Liang Xiuling, Li Peng, Chen Qingfei, et al. Retrieval practice benefits the cognitive neuroscience basis of learning and memory [J]. Psychological Science Progress, 2015, 23(7): 1151-1159.
[39] Myers K M, Davis M. Mechanisms of fear extinction [J]. Molecular psychiatry, 2007, 12(2): 120.
[40] Anderson M C. Rethinking interference theory: Executive control and the mechanisms of forgetting [J]. Journal of memory and language, 2003, 49(4): 415-445.
[41] Benoit R G, Anderson M C. Opposing mechanisms support the voluntary forgetting of unwanted memories [J]. Neuron, 2012, 76(2): 450-460.
[42] Anderson M C, Hanslmayr S. Neural mechanisms of motivated forgetting [J]. Trends in cognitive sciences, 2014, 18(6): 279-292.
[43] Wang Y, Cao Z, Zhu Z, et al. Cue-independent forgetting by intentional suppression–Evidence for inhibition as the mechanism of intentional forgetting [J]. Cognition, 2015, 143: 31-35.
Related Articles
Series 9 | Blue Book on Neuroscience and Education: How Attention in the Brain Promotes Learning (Part 2)
Series 8 | Blue Book on Neuroscience and Education: How Attention in the Brain Promotes Learning (Part 1)
Series 7 | Blue Book on Neuroscience and Education: General Principles of Brain Systems (Part 2)
Series 6 | Blue Book on Neuroscience and Education: General Principles of Brain Systems (Part 1)
Series 5 | Blue Book on Neuroscience and Education: Basic Structure and Function of the Brain (Part 2)
Series 4 | Blue Book on Neuroscience and Education: Basic Structure and Function of the Brain (Part 1)
Series 3 | Blue Book on Neuroscience and Education: How Far is it from Neuroscience to Educational Practice
Series 2 | Blue Book on Neuroscience and Education: Neuro Myths (Part 2)
Series 1 | Blue Book on Neuroscience and Education: Neuro Myths (Part 1)
Series Article Overview | Blue Book on Neuroscience and Education
