Click the blue text to follow us

The combination of PDT and PTT has proven to be a highly effective strategy, effectively compensating for their respective limitations and fully leveraging the advantages of combined therapy. ROS-mediated PDT can damage HSP, which helps improve the anti-tumor effects of PTT. On the other hand, PTT can ablate tumors regardless of oxygen levels. Additionally, it can enhance blood circulation through heat production, thereby increasing oxygen content in hypoxic tumor tissues, thus improving the therapeutic effect of PDT. Therefore, the shortcomings of PDT and PTT can be balanced through synergistic effects, achieving an effect of “1+1>2”. Moreover, non-targeted PSs have limited effectiveness in treating tumors and may cause severe side effects by damaging normal tissues. To improve cancer treatment, selectively delivering drugs to specific subcellular organelles is an effective strategy. Since mitochondria can provide energy to tumor cells, targeting mitochondria with PTAs has shown promising prospects for efficient anti-tumor therapy. Mitochondria also play significant roles in cell differentiation and metabolism, making them sensitive to high temperatures and oxidative damage. Therefore, developing PSs that can target tumor cell mitochondria and possess efficient I-type photodynamic and high PCE is crucial for multimodal synergistic diagnosis and treatment of tumors. The related paper, πBridge Engineering-Boosted Dual Enhancement of Type‑I Photodynamic and Photothermal Performance for Mitochondria-Targeting Multimodal Phototheranostics of Tumor, was published on November 1, 2023 in ACS Nano (IF=17.1).
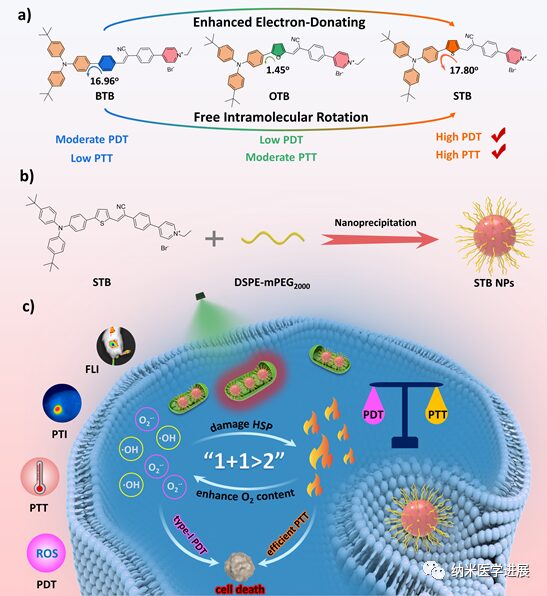
Figure 1: Cationic photosensitizer STB for multimodal therapy targeting tumor cell mitochondria
This paper employs the π bridge engineering strategy to construct a series of cationic PSs with a D-π-A structure for the synergistic treatment of I-type PDT and PTT. (1) The cationic pyridine groups can actively target the mitochondria of tumor cells through electrostatic interactions; (2) By modulating the electron-donating ability of the π bridge, STB exhibits red-shifted absorption and emission, with an emission peak tailing over 1000 nm in the solid state; (3) The stronger ICT and smaller ΔES-T of STB promote ISC and generate a large amount of O2•– and •OH for I-type PDT; (4) The high molar extinction coefficient and more free intramolecular rotation of STB effectively promote photothermal conversion for efficient PTT. In vitro and in vivo experiments indicate that STB NPs can effectively inhibit cell proliferation and tumor growth through NIR-FLI and PTI-guided I-type PDT and PTT synergistic therapy.
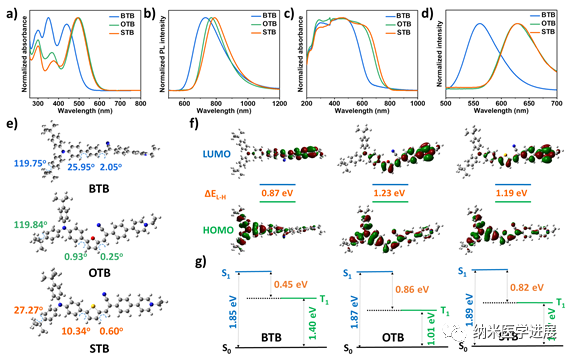 Figure 2: Spectral properties and theoretical calculations of the photosensitizer
Figure 2: Spectral properties and theoretical calculations of the photosensitizer
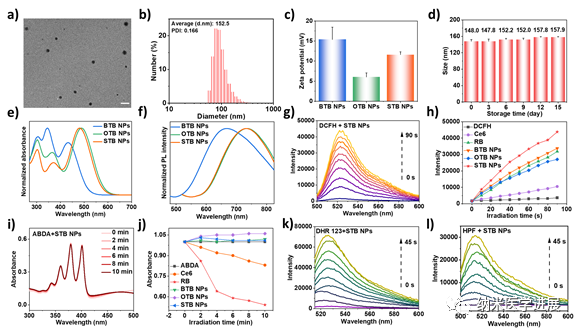 Figure 3: Fundamental properties and photodynamic performance characterization of nanoparticles
Figure 3: Fundamental properties and photodynamic performance characterization of nanoparticles
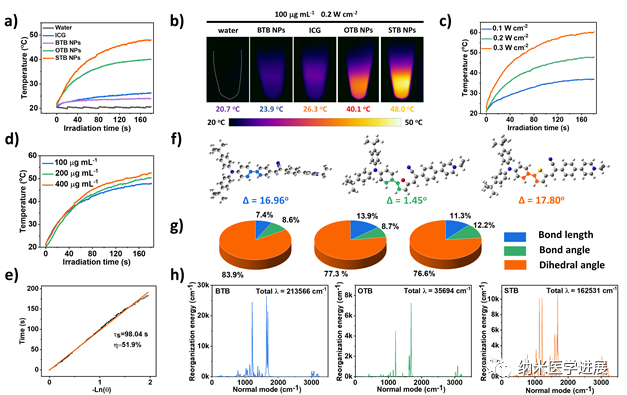 Figure 4: Photothermal performance of nanoparticles
Figure 4: Photothermal performance of nanoparticles
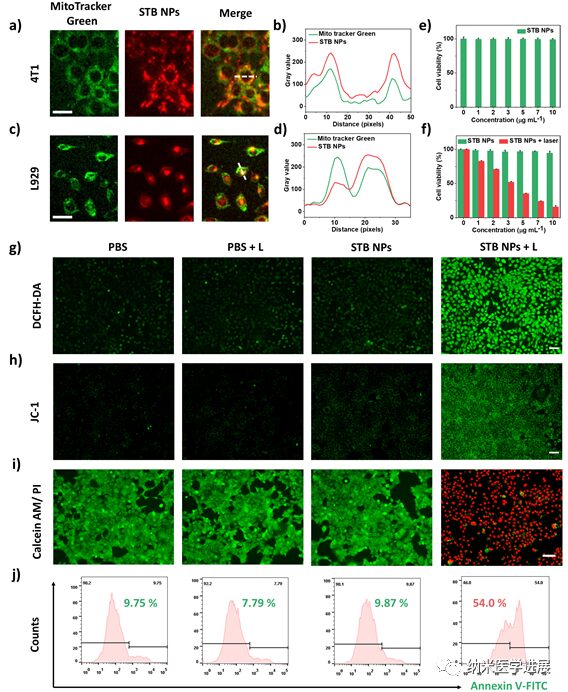 Figure 5: Targeting, biocompatibility, and phototoxicity of STB NPs at the cellular level
Figure 5: Targeting, biocompatibility, and phototoxicity of STB NPs at the cellular level
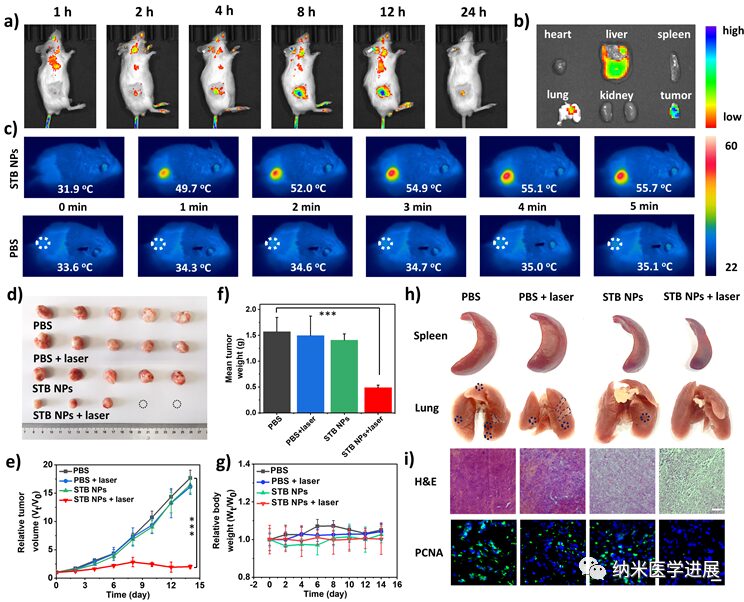 Figure 6: Antitumor therapeutic performance of STB NPs in vivo
Figure 6: Antitumor therapeutic performance of STB NPs in vivo
This paper cleverly designs and synthesizes a series of phototherapeutic agents with a D-π-A structure that can target tumor cell mitochondria. By adjusting the electron-donating ability of the π bridge, the STB with stronger D-A interactions, smaller ΔEST, and sufficient intramolecular motion achieves optimal I-type ROS generation and a high photothermal conversion efficiency of 51.9% under laser irradiation. STB NPs perform well both in vitro and in vivo, actively targeting and accumulating in tumor regions, enabling real-time NIR fluorescence imaging and significant antitumor activity with low side effects. This work not only provides new perspectives for designing phototherapeutic agents with balanced PDT and PTT performance but also promotes the development of integrated platforms for tumor diagnosis and treatment guided by multimodal imaging.
Paper link: https://pubs.acs.org/doi/10.1021/acsnano.3c06542
Disclaimer: The content shared by this WeChat account is for academic exchange only and does not serve commercial purposes, nor does it represent support or endorsement of its views. If there are issues related to intellectual property protection or other matters, please contact the editor in a timely manner, and we will coordinate to handle it as soon as possible. The copyright of original articles from this WeChat account belongs to this WeChat account, and sharing on personal social media is welcome.
This public account also has a real-name academic exchange group for nanomedicine. Please add the WeChat ID below with your name + organization + research direction.